Key Takeaways
- Peptides are essentially short strings of amino acids that act as biological messengers and can be leveraged to support everything from tissue repair to metabolism to immune function to targeted therapies. Think about getting an option with a competent clinician prior to starting any protocol.
- Peptide therapies have the potential to optimize athleticism, body composition, recovery, and even anti-aging by manipulating protein production and hormonal pathways. There are some quantifiable metrics, such as muscle mass, fat percentage, and recovery times, to gauge impact.
- Personalized approaches matter because genes, biomarkers, health history, and current medications alter how individuals respond to peptides. Get genetic or biomarker testing and adjust dosages and combinations accordingly.
- Risks such as side effects, immunogenic reactions, and low quality sources minimize risk with pharmaceutical-grade peptides, verified suppliers, and regulated healthcare providers.
- About how societal expectations will shift alongside peptide enhanced bodies – about health, appearance, and longevity, as well as access, fairness, and ethics. Watch policy, insurance, and clinical guidelines as access and norms change.
- Down the line we should see an expansion of peptide types, delivery methods, and combination therapies. Stay on top of the clinical data and make a point of looking for safety, efficacy, and individual benefit before embracing new options.
What expectations will change with peptide enhanced bodies that social and medical norms will shift toward performance and recovery metrics?
We will judge strength, healing time, and appearance not by how you describe it but by muscle gain rates, wound recovery days, and hormone balances. Employers, insurers, and sports groups will rework policies to new baselines.
The body concerns ethical, legal, and health implications and probable adoption timelines.
Understanding Peptides
Peptides are mini-proteins that serve as biological messengers. They regulate growth, repair, immunity, and hormone signaling. The body has 20 common amino acids to craft thousands of different peptide sequences, each serving a distinct function. Some mimic natural hormones, while others induce cells to produce more hormones.
Understanding what peptides do and how they act in the body is critical to estimating how expectations will change when people are using peptide-augmented bodies.
The Science
Peptides bind to particular receptors on the surfaces of cells. That binding in turn triggers cascades that change gene expression, protein synthesis, or metabolic activity. For tissue repair, for example, peptides can tell fibroblasts and stem cells to reproduce and deposit extracellular matrix.
For metabolism, they modify routes that alter how cells consume glucose and fat. Delivery counts. Injected peptides drain immediately into the bloodstream or local tissue, providing predictable exposure. Oral peptides tend to degrade in the gut, with many having as little as 1 to 2 percent bioavailability.
Some, like collagen peptides, can achieve significantly greater oral bioavailability, with reports as high as around 63 percent, and some small peptides like GHK-Cu can permeate skin upon topical application. Storage and handling influence activity. The majority of peptides remain stable if stored at or around 4 degrees Celsius (39 degrees Fahrenheit).
Endogenous production is not the same as synthetic dosing. Peptides that are endogenous are released in time-limited bursts and cleared rapidly. Synthetic administration can maintain higher levels, extending effects or pushing responses outside natural boundaries.
That can boost advantages, but it shifts risk profiles and impacts feedback on hormone axes. Peptide pathways enhance immunity, muscle development, and cellular longevity by activating repair and anti-inflammatory pathways. These pathways illuminate why peptides are increasingly interesting across wellness and medicine.
The Types
Metabolic peptides govern energy usage and body composition. Immune peptides sculpt inflammation and pathogen defense. Antimicrobial peptides assault microbes and promote barrier health. Neuropeptides change mood, pain, and behavior.
Injectables are more suited for focused, powerful impact such as growth hormone secretagogues for muscle and recovery. Oral supplements fit systemic, lower-dose aims like collagen for skin and joints. Topical peptides can assist skin repair when the molecular size allows penetration.
| Peptide Type | Primary Function | Common Therapeutic Uses |
|---|---|---|
| Metabolic peptides | Regulate metabolism, appetite | Weight management, GH release |
| Immune peptides | Modulate inflammation | Autoimmunity, infection support |
| Antimicrobial peptides | Kill microbes, heal barriers | Wound care, topical infection control |
| Neuropeptides | Signal in nervous system | Pain, mood, cognition support |
The Distinction
Peptides tend to be smaller than proteins and serve as signals, not structures. Peptide hormones tend to bind to surface receptors and generate rapid, targeted actions. The larger protein hormones can play more widespread, enduring roles.
In comparison to traditional therapeutics, peptides can be more target-selective with minimal off-target effects but may face stability challenges and necessitate specialized formulation and administration. These unique mechanisms, including receptor specificity, short half-lives, and pathway-focused action, make peptides useful for a variety of targeted wellness approaches.
Redefining Physical Limits
Peptide therapies are redefining physical limits by altering the equilibrium between organic potential and augmented optimization. They function on metabolism, tissue repair, and signaling. Here’s a brief look at the key areas where expectations will shift and what that means in practice.
1. Athletic Performance
Growth hormone–releasing peptides and metabolic peptides are able to raise lean mass and improve endurance by inspiring protein synthesis and increasing mitochondrial efficiency. Athletes utilize peptides such as GHRP-6, ipamorelin, and BPC-157 to maintain muscle hypertrophy and recovery after intense sessions.
These peptides assist in maintaining high training volume without experiencing chronic fatigue. Peptide injection protocols shave standard recuperation time frames. By accelerating the repair of microtears, downtime between brutal workouts is minimized and adaptation cycles are shorter, allowing athletes to sustain elevated training loads on a weekly basis.
Some peptides further blunt exercise-induced inflammation, which reduces delayed onset muscle soreness and supports consistent performance. Certain peptides, like TB-500 and BPC-157, pop up in user reports for tendon and soft-tissue resilience. A practical list for performance-minded users might include: ipamorelin (growth support), BPC-157 (soft-tissue repair), TB-500 (cell migration), and CJC-1295 (GH pulse enhancement).
Protocols vary by sport and objective. Endurance athletes prefer mitochondrial and metabolic modulators. Strength athletes favor GH secretagogues.
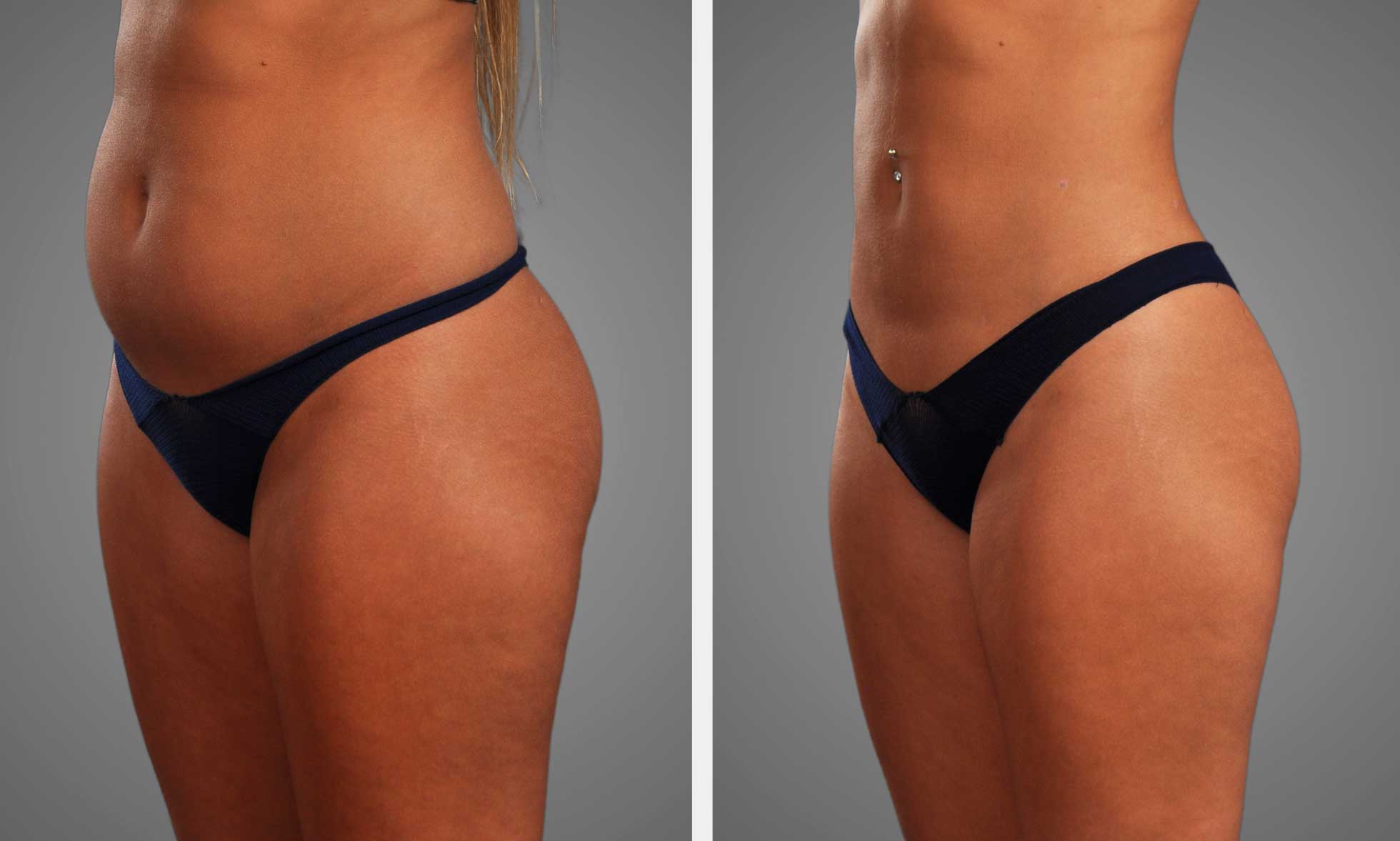
2. Body Composition
Some peptides turn fuel utilization toward fat and maintain muscle during calorie deficits. GLP-1 receptor agonists and their analogues slow gastric emptying, reduce appetite, and increase insulin sensitivity, which facilitates consistent fat loss with minimal muscle loss when accompanied by resistance training.
Some of the other peptides help preserve muscle mass in an aging or dieting state by supporting anabolic signaling. For those eating fewer calories, supplementing with a muscle-sparing peptide will stop strength losses and preserve metabolic rate.
Trackable metrics, such as fat percentage, lean mass (DXA), waist circumference, and fasting insulin, demonstrate significant alteration.
3. Injury Recovery
Peptides can accelerate tissue repair, helping with ligament healing and muscle regeneration. Collagen peptides provide raw materials for connective tissue, while thymosin beta-4 and BPC-157 regulate repair cascades and minimize scars.
Chronic wounds respond in some cases to targeted peptide protocols that enhance local blood flow and cell turnover. Tailor peptide selection to injury and anticipated time frame. Tendinopathy might require a different strategy than a muscle strain.
Clinicians often bullet point options and anticipated recovery weeks in order to facilitate patient scheduling.
4. Anti-Aging
Longevity peptides and GH secretagogues seek to decelerate biological aging by reducing chronic inflammation, promoting skin regeneration, and enhancing mitochondrial function. Peptides such as sermorelin and epithalon are studied for telomere effects and sleep regulation.
Neuroprotective peptides, such as certain BDNF fragment modulators, aid in cognitive resilience and neuron repair. A quick reference table of peptides, doses, and effects guides clinicians in selecting protocols for specific anti-aging objectives.
5. Everyday Wellness
Peptide regimens can boost baseline energy, bolster immune responses, and stabilize metabolic regulation. Immune peptides aid quicker recuperation from common sickness and metabolic peptides help stabilize blood sugar and hunger.
Integrate peptides into routines with clear goals: sleep quality, energy, or immune resilience. Define clear, achievable results and check them after specified periods to adapt your treatment.
The Personalization Factor
Peptide therapy is most effective when it’s a good fit for the individual. The personalization factor in personalized medicine for peptide use is about constructing regimens from an individual’s complete health profile — genetics, biomarkers, medications, lifestyle, and objectives. That minimizes trial and error, decreases side effects, and increases the probability of long-term benefit for conditions like inflammation, weight management, and insulin resistance.
Genetic Markers
Genetic testing helps select who will likely respond to certain peptides and at what dose. Polymorphisms in genes can affect peptide uptake, receptor sensitivity and downstream metabolism, so the same peptide can act fast in one person and slow in another. Tests identify risks, like variants impacting clotting, liver enzymes or hormone pathways, which informs safe selection of compounds and microdosing when necessary.
- APOE (lipid and inflammation responses)
- MTHFR (methylation, detox, and metabolic status)
- FTO (weight regulation tendencies)
- COMT (neurotransmitter breakdown, stress response)
- CYP450 family (drug and peptide metabolism)
- IGF1/IGF1R (growth factor sensitivity)
- LEPR (leptin receptor, appetite control)
These markers are routinely tested to personalize peptide choice, dosages, and synergies with nutrition or other medications.
Biomarker Testing
Blood and saliva tests monitor peptides’ impact on hormones, metabolic markers and organ health. As a prelude, baseline measures create goals. While in therapy, serial testing reveals if peptides alter insulin sensitivity, inflammatory cytokines, lipid panels or liver enzymes. Tracking regularly helps you catch undesired shifts early and verify benefit.
Key biomarkers checklist:
- Fasting glucose and HbA1c
- Insulin and C‑peptide levels
- CRP and ESR (inflammation)
- Lipid panel (HDL, LDL, triglycerides)
- Liver enzymes (ALT, AST)
- Thyroid panel (TSH, free T4, free T3)
- IGF‑1 and relevant hormones
- Vitamin D and basic micronutrients
Tests at baseline, 4 to 12 weeks after starting, then every 3 to 6 months depending on therapy intensity.
Tailored Protocols
Tailored protocols start with a full medical review: history, current meds, allergies, and lifestyle habits like sleep, diet, and exercise. They select peptides and doses by goals, which include reducing inflammation, improving body composition, or enhancing metabolic flexibility, along with raw data from genes and biomarkers.
Microdosing is used for sensitive people to reduce side effects and build benefits gradually. Continuous attention translates to regular check-ins and dose adjustments. Recording results, such as symptom scores, lab trends, and side effects, makes future plans more intelligent.
Integrate peptides with supportive practices, including meal planning for metabolic goals, controlled exercise for muscle response, and stress management to improve hormonal balance. The outcome is a strategy that shifts with the individual, not a cookie-cutter regimen.
Navigating The Risks
Peptide-enhanced bodies come with medical promise and practical risks. Before they begin treatment, readers require a transparent perspective on the probable harms, where the risk emerges, and how to mitigate those risks. These points encompass side effects, sourcing risks, and regulatory challenges. They illustrate why medical supervision and proven alternatives are important.
Side Effects
Typical acute side effects are local pain, erythema, and swelling. Systemic reactions may manifest as fever, fatigue, or allergies. Other peptides initiate immune responses that alter the body’s response to subsequent exposure, which can cause loss of effect or more serious autoimmune issues.
Beyond mild allergic reactions, some peptides have been associated with fatal outcomes. It’s been implicated in causing other issues like reported nausea, kidney stress or failure, skin flushing, and even potential links to skin cancer and cardiovascular strain. GLP-1 receptor agonists, for instance, can alter appetite and metabolism and may be especially dangerous for women during midlife or menopause.

Evidence for benefits varies. Ipamorelin remains mostly promise rather than proof, and microdosing strategies lack evidence and raise long-term safety questions. Just a thought – why not compile a handy clinic-side reference table of peptide side effects and frequency and use it for patient consent? This allows clinicians and patients to balance particular risks by compound and by dosage.
Sourcing Dangers
Purchasing peptides from unauthorized dealers presents a significant danger. These low-grade suppliers can peddle products with impurities, contamination, or inappropriate dosing, risks that directly impact safety and efficacy. A few peptides, including GHRP-2, GHRP-6, and BPC-157 were banned from compounding due to safety and data concerns. Offering banned items is a red flag.
Pharmaceutical-grade peptides with third-party purity testing require checking for certificates of analysis from credible labs and requesting batch testing results. Proper storage and handling matter. Many peptides need cold-chain storage and must be reconstituted and dosed precisely. Clinics must reject undocumented products or those that come in inappropriate packaging.
Regulatory Hurdles
They are where regulators mold access and safety. The FDA and its international equivalents create boundaries for clinical use, approvals, and marketing, with many experimental peptides still off-label or unapproved. This builds walls to legal access and results in clinical data being sparse.
It faces challenges such as a scarcity of human studies, particularly those involving women or older adults. Legal status varies by jurisdiction, with some peptides banned or controlled in compounding. Current hurdles include sparse clinical evidence, uneven regulation across countries, restrictions on compounding certain peptides, and slow pathways for proof of long-term safety.
Prioritize proven therapies, use comprehensive clinical services, and inquire about human studies before initiating treatment.
The Societal Shift
These peptide-optimized bodies are redefining what individuals expect from health and beauty by shifting it away from disease management and toward performance, recovery, and continuous maintenance. With over 100 peptide drugs approved by the FDA in the US and the global peptide market exceeding approximately 70 billion USD in 2019, the tech is transitioning from niche clinics to mainstream care.
This part unpacks how those shifts manifest in norms, demand, perceptions, and numbers.
Body Image
Peptides that accelerate healing or transform body composition or skin quality shift the baseline for what is considered achievable. Quicker healing from injury and noticeable improvements in muscle tone or fat reduction can reset standards. Folks begin to anticipate less obvious development or faster recovery to pre-injury fitness.
The surge in aesthetic applications, such as skin rejuvenation and weight-loss regimens, is already transforming standards in cities and elsewhere. Pressure can follow. When treatments shorten the time between effort and visible change, individuals may feel compelled to pursue peptides to stay competitive socially or professionally.
Psychological risks include altered self-worth, body dysmorphia, and therapy dependence. Clinicians and clinics should evaluate motivation, define achievable goals, and provide mental health screening prior to aesthetic protocols.
Accessibility
Access to peptide therapies remains uneven, influenced by factors such as cost, geographic concentration of clinics specializing in peptide treatments, and differences in healthcare systems globally. Out-of-pocket costs and networks of clinics make early adopters more likely to be wealthier or urban.
Ambitious attempts to expand access encompass telemedicine consults, scaled clinic models, and pilot public-health initiatives that fold regenerative approaches into routine care. Tracking insurance coverage changes and public policy shifts is essential.
Expanding coverage would alter who can obtain peptides for chronic inflammatory conditions or age-related decline. Wellness clinics utilizing both remote monitoring and localized dosing can decrease travel burdens, but infrastructure and trained workforce are still barriers in many regions.
Ethical Lines
Peptide usage brings up obvious ethical dilemmas around fairness, consent, and intention. Peptides to help an athlete break records or heal faster than what is naturally achievable or look better is unfair and confuses anti-doping regulations.
Informed consent has to address off-label usage, different individual responses, and long-term unknowns. Personalized medicine is increasingly critical as patients respond differently to peptide protocols.
Regulators and professional bodies require transparency on therapeutic versus enhancement use, disclosure standards, and monitoring. Key ethical questions include who decides acceptable uses, how to prevent coercion, and how to ensure equity in access and oversight.
Future Trajectory
Peptide-enhanced bodies will transfer expectations as analysis, delivery and medical observe into the future. The market is expanding from approximately $25.3 billion in 2024 to an anticipated $41.7 billion by 2030, with over 200 peptides undergoing clinical development. That scale of investment and pipeline work implies more rapid innovation around novel peptide discovery, clearer use cases in metabolic, immune and regenerative health, and more advanced chemical modifications to improve the stability and specificity of peptides.
Forecast for peptide research, new peptides and new treatments. New peptides will be designed with particular targets in mind, minimizing side effects and increasing potency. Peptides will be designed to imitate hormone signals for appetite regulation or peptides that attach to inflammatory receptors to stem persistent tissue damage. Enhanced stability, including extended half-lives and resistance to enzymes, will require lower doses and fewer administrations.
Oral formulations and slow-release implants will cross from lab proof-of-concepts to real-world alternatives, transforming the perception of peptide therapy from clinic-only jabs to at-home regimens. Peptides will carve out a space for themselves between mainstream medicine and personalized wellness. Peptides will be more often standard care, not fringe.
Clinicians will use peptide panels in combination with genetics, biomarkers, and imaging to construct treatment plans. For instance, an individual with early insulin resistance could receive a peptide that enhances glucose processing combined with nutrition guidance and regular testing. Regulatory pathways, funding, and public education will guide pace and extent. In jurisdictions with explicit coverage and reimbursement, adoption will be faster. Where regulations are lagging, adoption should be slower.
Emphasize the possibilities of combinatorial therapies and synergistic longevity effects. Peptides will combine with small molecules, biologics, and lifestyle. Pairing a peptide that reduces inflammation with one that supports mitochondrial function, for example, may provide more benefit than either by itself.
In aging research, combinatorial strategies could decelerate several aging mechanisms such as metabolic regulation, cellular maintenance, and immune homeostasis, providing more expansive functional advantage. They will require clinical trials that are designed to test combinations and capture long-term outcomes rather than single biomarkers.
Suggest keeping an eye on industry trends and innovations in peptide therapy for continuous improvement. Watch for delivery innovations, such as oral and transdermal, among others, news candidates targeting obesity, diabetes, and complex chronic diseases. Follow clinical trial results for the 200 plus peptides in development, regulatory guidance changes, and pricing shifts driving market growth.
These individual plans will need regular re-evaluation as new options arise so that clinicians and well-informed patients can optimize safety, cost, and outcomes.
Conclusion
Peptides will change what we expect from bodies. With short recovery times, more steady gains in muscle, and clearer skin, higher performance will feel normal. Athletes, weekend warriors, and rehab patients will select targets that fit their lifestyle and objectives. Health pros will hone in on tests, dose plans, and side-effect checks to keep results safe and steady. For society, fair play, cost, and access will be daily decisions. Policy and sport rules will adjust to new norms. Some transparent, easy steps in oversight, education, and care will make it possible for people to benefit without harm.
If you want a no-nonsense guide to safe peptide use, a test-driven plan, or a quick vetting of the hottest peptides, DM and I will outline options.
Frequently Asked Questions
What are peptide-enhanced bodies?
Peptide-enhanced bodies manipulate biology with peptides — little chains of protein. They can assist with muscle growth, recovery, fat burning, or healing. Medical oversight and reputable sources are critical to safety and efficacy.
How will expectations for athletic performance change?
Expectations will increase for rapid recovery and more efficient muscle gains. Athletes would achieve greater training volumes earlier. Results differ by peptide type, dosage, and personal biology.
Will personalized treatments become the norm?
Yes. Precision dosing based on genetics, biomarkers, and goals will drive better results. Customization minimizes adverse effects and optimizes the benefit to risk ratio.
What are the main health risks?
Risks include dosing errors, contamination, immune responses, and uncharted long-term consequences. Unregulated products can cause harm. Clinical guidance mitigates these risks.
How will society view body standards?
Expectations might transition to performance and functionality rather than looks. This may access disparities, widen social gaps, and create ethical and regulatory debates about fairness.
Could peptide use affect workplace or insurance policies?
Employers and insurers might revise performance enhancement, safety screening, and coverage policies. Legal and ethical frameworks will shift with medical evidence.
What is the likely future of peptide technology?
Anticipate safer and more targeted peptides, better delivery methods, and stronger regulations. Wider clinical trials will illuminate advantages and hazards and direct cautious embrace.