Key Takeaways
- Retatrutide provides triple hormone receptor activation: GLP-1, GIP, and glucagon to support blood sugar, appetite, and fat metabolism together.
- Its triple receptor agonism demonstrates potential benefits for enhanced weight loss and metabolic health, providing benefits over conventional single-receptor therapies.
- Clinical trials indicated that patients treated with retatrutide experienced significant weight loss, better glycemic control, and reduced liver fat.
- Retatrutide’s innovative peptide design drives effective receptor targeting that leads to improved metabolic outcomes and durability.
- Safety data indicate retatrutide is typically well-tolerated. Continued vigilance is essential to support safe administration across heterogeneous cohorts.
- Retatrutide could potentially tackle numerous obesity-related issues from cardiovascular to renal, positioning itself as a noteworthy contender in the upcoming metabolic disease arsenal.
Retatrutide triple receptor activation means the drug works by binding to three key hormone receptors in the body: GLP-1, GIP, and glucagon. All of these receptors help regulate blood sugar, appetite, and metabolism.
By working on all three, retatrutide hopes to provide a more powerful impact for weight loss and diabetes management. If you want to understand how this works in detail, the body breaks down each receptor and its role.
Triple-Agonist Mechanism
Retatrutide is a triple hormone receptor agonist. It activates three main receptors: GLP-1, GIP, and glucagon. All three have a different role in glucose homeostasis and metabolism. It is designed to sequentially target multiple metabolic pathways simultaneously, which can aid obesity, liver health, and cardiac risks.
1. GLP-1 Receptor
GLP-1 receptor prompts the pancreas to produce insulin when blood sugar rises after meals. It delays gastric emptying and induces satiety, so you consume less. We know GLP-1 activation lowers blood glucose by increasing insulin and decreasing glucagon. This can assist individuals in maintaining their blood sugar in a healthier range from day to day.
Research on retatrutide indicates that its GLP-1 effect is a significant factor behind the impressive weight loss experienced, exceeding 20% body weight reduction in numerous patients. Lower blood sugar and less hunger in combination result in improved metabolic health and decreased risk for diabetes.
2. GIP Receptor
GIP is glucose-dependent insulinotropic polypeptide. This receptor increases insulin secretion post-prandially, depending on glucose levels. GIP activation contributes to fat storage and energy expenditure.
With retatrutide activating GIP, individuals might torch additional fat and achieve enhanced regulation of their energy storage and utilization. GIP’s impact on metabolism contrasts with GLP-1, as it could maintain energy homeostasis without risking hypoglycemia.
In obesity, GIP receptor activation is believed to contribute to the weight loss and metabolic effects observed with triple-agonist agents.
3. Glucagon Receptor
Glucagon receptor primarily acts on the liver, instructing it to produce more glucose when blood sugar is low. In obese or diabetic individuals, this can be an issue. Balanced activation can assist in fat burning and reducing liver fat.
Retatrutide’s action on the glucagon receptor means less liver fat, with up to 86% reduction in liver fat observed in trials. Targeting this receptor can aid in managing appetite, as glucagon has the ability to reduce hunger in certain instances.
This action assists in liberating stored fats for fuel, contributing to the loss of weight and waistlines.
4. Synergistic Action
When GLP-1, GIP, and glucagon receptors are simultaneously activated, their effects are synergistic. This triple action aids insulin sensitivity, appetite control, and fat loss. The effect is more weight loss than you get with drugs that hit only one receptor.
Retatrutide’s mechanism supports heart and liver health by decreasing fat stores in vital areas such as the liver and surrounding organs. Weekly injections are possible because of its long half-life, and its time-staggered effects on the different pathways enable a steadier, more predictable response with fewer sugar or energy highs and lows.
Such a balanced approach has extensive advantages in individuals with obesity or metabolic disease.
Molecular Design
Retatrutide, in particular, is a synthetic peptide designed to activate several receptors simultaneously. At its heart, it’s designed to bind GLP-1, GIP, and glucagon receptors. This triple receptor activation distinguishes it from older medications that target only one or two.
Molecular design applies both in silico and structure-based approaches to create novel peptides with defined biological activities. These approaches allow researchers to select the proper amino acid sequence and construct elements such as α-helices that can conform tightly to their destined receptors. In retatrutide’s case, the sequence and shape are selected to bind and activate all three receptors.
It can act on multiple pathways in the body, which is why it could be helpful for complicated diseases such as type 2 diabetes and obesity.
Peptide design isn’t only about selecting the correct sequence. It’s about making the molecule stable and lasting in the body. For peptides such as retatrutide, attaching fatty acyl groups, known as acylation, assists the drug in remaining in the bloodstream for an extended period of time.
This approach was applied previously in drugs like semaglutide and tirzepatide. This fatty acid ‘tail’ allows the peptide to bind to proteins in the bloodstream, which delays breakdown and enables less frequent dosing. This is crucial for patient convenience and maintaining stable blood levels.
The folding of the peptide, usually as an α-helix, additionally assists it in maintaining its shape and binding with high affinity to its targets. Through molecular design, researchers can tune these characteristics to strike a compromise between potent binding, durability, and safety.
Retatrutide’s pharmacokinetics describe its journey through the body. Retatrutide is formulated for subcutaneous injection, and its acylated structure facilitates a long half-life. This allows for once a week dosing, like other newer GLP-1s.
These sustained blood levels help the medicine continue working and reduce any fluctuations in effectiveness that may occur with shorter-acting medications. A long half-life means less risk of forgetting a dose or experiencing huge fluctuations in effect. Dosing regimens are constructed around these characteristics, simplifying adherence.
- Key advancements in peptide technology for triple receptor agonism:
- Application of computational modeling to peptide-receptor binding
- Fatty acid substitution for extended stability and duration
- α-helical design for superior receptor fit
- Structure-based design to hone drug action and safety
- Engineering peptides that simultaneously bind and stimulate multiple receptors
Clinical Implications
With retatrutide, a triple receptor agonist that has been studied in clinical trials for obesity and metabolic diseases, including type 2 diabetes and MASLD. Clinical experience underscores the potential and subtlety of its impact.
| Outcome | Observation/Value | Timeline | Safety Notes |
|---|---|---|---|
| Mean weight loss | Up to 24% | 48 weeks | Most AEs mild/moderate |
| Liver fat reduction | Up to 76% | 24–48 weeks | GI symptoms most common |
| VAT reduction | 12.9%–43.5% | 24–48 weeks | No severe hypoglycemia |
| Insulin resistance markers | As much as 50% less | 24 to 48 weeks | No major cardiac events | Triglyceride reduction | Large, correlated with liver fat | 24 to 48 weeks |
Weight Loss
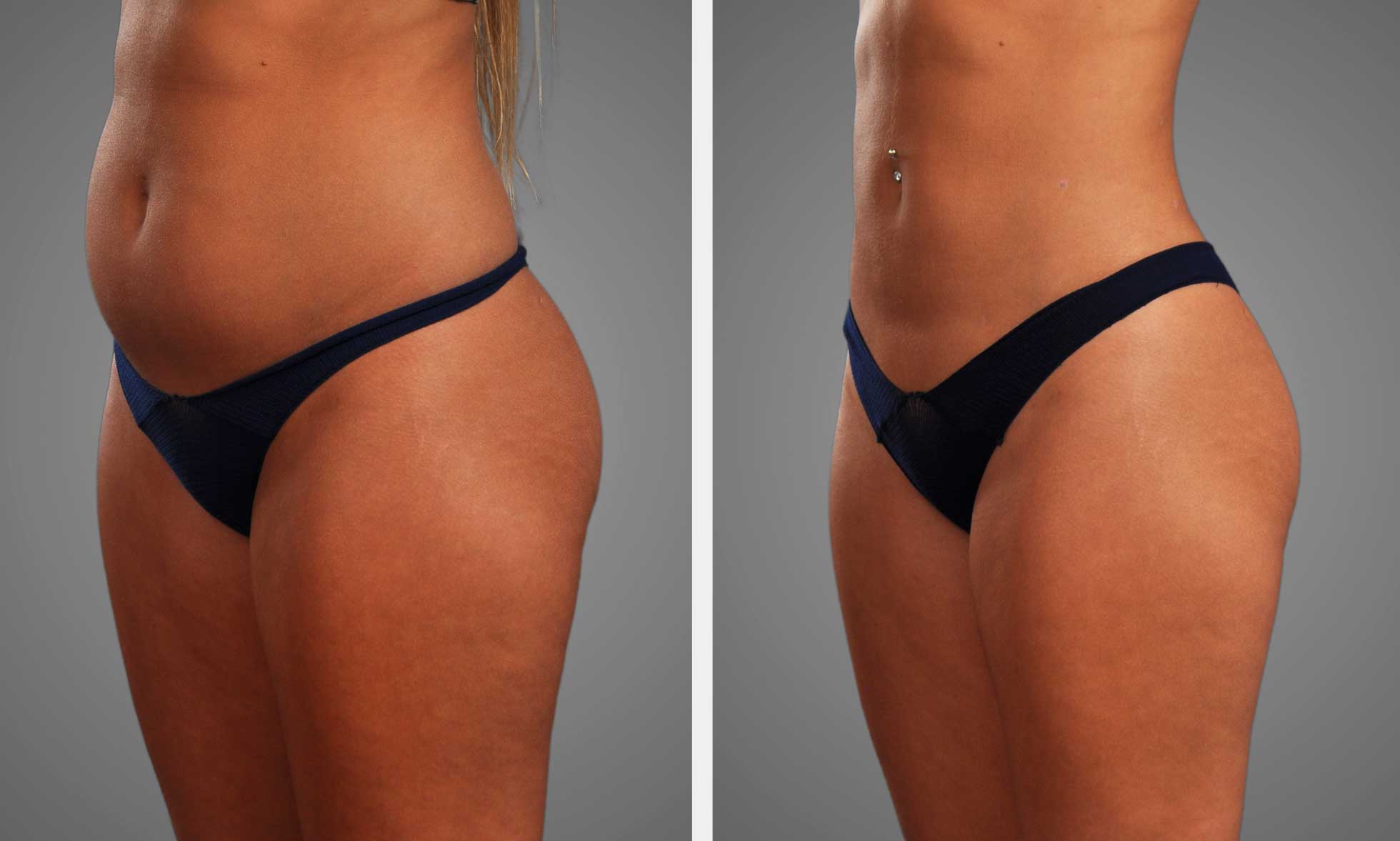
Retatrutide resulted in significant weight loss in multiple clinical trials. Participants experienced as much as 24 percent body weight loss at 48 weeks at higher doses. This degree of weight loss exceeds most existing medications, including single or dual agonists.
Initial information indicates a majority of the weight loss occurs within the first 24 weeks, demonstrating a rapid time to onset. The triple receptor activation hits multiple pathways. By targeting GLP-1, GIP, and glucagon receptors, retatrutide suppresses appetite, delays gastric emptying, and increases energy expenditure.
Together, these effects support sustained weight loss, as opposed to single mechanism drugs. The majority of the lost body fat was visceral and abdominal. VAT and ASAT decreased by up to 43.5 percent after 48 weeks.
Appetite control appears to be at the center of sustained weight loss with retatrutide. Most users find themselves less hungry and more satiated. This facilitates calorie restriction and promotes long-term compliance.
Obese adults, individuals with type 2 diabetes, and MASLD may derive the most advantage. Trials enrolled men and women, and benefits were evident across different ethnicities and age groups.
Metabolic Health
Clinical Implications: While human trials are limited, they demonstrate significant metabolic improvements particularly with respect to insulin resistance and glucose control. Fasting insulin, C-peptide, and HOMA2-IR decreased by 50% or more from baseline. These modifications can reduce the risk of diabetes-related complications.
Retatrutide improves insulin sensitivity and stabilizes blood sugar. This is crucial for individuals with prediabetes or type 2 diabetes. Markers such as triglycerides, adiponectin, leptin, and FGF21 were enhanced and these tracked with liver fat reduction.
By tackling multiple components of metabolic syndrome, such as adiposity, blood glucose, and lipid profiles, retatrutide provides a more holistic strategy than earlier medications.
- Lower fasting insulin and C-peptide
- Reduced HOMA2-IR (insulin resistance)
- Improved lipid profile (triglycerides)
- Raised adiponectin, lowered leptin
- Better glucose control
Liver Fat
Retatrutide reduced liver fat content by as much as 76% for MASLD patients, with the majority of the change occurring during the first 24 weeks. This fall in liver fat is strongly associated with body weight loss, with close to maximal liver fat change occurring at approximately 20% total weight loss.
Lowering liver fat is important in managing or preventing progressive liver disease. Less liver fat means less risk of fibrosis, cirrhosis, and other complications. It enhanced liver function markers, indicating improved liver health.
The connection between liver fat reduction and decreased insulin, C-peptide, triglycerides, and increased adiponectin suggests there is a wider metabolic benefit, not just localized liver effects. Early and robust liver fat loss suggests that initiating treatment soon after diagnosis may yield optimal results.
Safety Profile
Retatrutide is a triple receptor agonist now being investigated for obesity, T2DM, and non-alcoholic fatty liver disease. Its safety profile has garnered attention as more individuals seek alternative ways to handle these conditions. Retatrutide maintains a good safety profile in phase 2 trials, like other GLP-1 receptor agonists. Researchers haven’t uncovered any major safety issues in preclinical studies, reinforcing early optimism.
The complete long-term safety landscape is not yet fully illuminated, which is why those continuing trials are key. Retatrutide is similar to OXM, which has been shown to improve insulin sensitivity, increase energy expenditure, and reduce food intake in obesity. These side effects are typical of other drugs in this class. For the majority of patients, these symptoms are mild to moderate and abate with time or dose adjustments.
There have been a couple of incidents of diarrhea or constipation, but severe events are uncommon. Physicians should observe for these adverse effects, notably in patients with comorbidities or polypharmacology. Careful dose steps and close follow-up can minimize the bother these side effects cause. Continued monitoring during clinical trials is essential for retatrutide’s safety.
The phase 3 trials now underway will help demonstrate how the drug performs over longer periods and in larger, more diverse populations of patients. This encompasses individuals with obesity, T2DM, and NAFLD, many of whom can have multiple comorbidities. Safety checks in trials include monitoring side effects, lab tests, and other health indicators to ensure that no new or rare issues emerge.
This vigilant strategy aids in identifying dangers early and maintains patient care secure. They haven’t given public-use approval yet for retatrutide. Its status remains controlled clinical trials, with use directed by stringent protocols and tight safety monitoring. The results of these ongoing studies will inform future approval decisions.
Once approved down the road, the safety profile observed in trials will guide real-world use of the drug, who can take it, and what additional measures might be necessary to protect patients.
Beyond Weight Loss
There’s more to retatrutide’s triple receptor activation than weight loss. Research reveals its impact extends well beyond weight loss to many aspects of the body’s metabolism. Many of our patients experience changes in markers related to liver health, blood sugar, and hormones. This renders it more than just a weight-control drug.
Retatrutide promises to help the body’s metabolism work better. It reduced liver fat by approximately 75% in clinical trials when individuals lost approximately 20% of their body weight. Post-treatment, over 80% of patients had healthy liver fat levels of less than 5%. This matters because elevated liver fat frequently causes other health issues.
The connection between decreased liver fat and changes in β-hydroxybutyrate at 24 weeks implies retatrutide could assist the body in burning fat more efficiently for fuel. Retatrutide reduced leptin levels at 24 and 48 weeks at specific dose levels. Lower leptin might help with hunger and fat storage, which is an extra bonus beyond weight loss.
Adiponectin, a separate hormone that aids in blood sugar and fat usage, increased at weeks 24 and 48 at doses of 4 mg or higher. This flip indicates improved metabolic fitness. FGF21, a protein implicated in fat and sugar utilization, decreased at higher doses, which may indicate enhanced metabolism. Pro-C3, a marker for liver scarring, decreased at multiple doses, suggesting improved liver health over time.
Another feature setting retatrutide apart is how it improves heart and kidney health. Type 2 diabetes patients experienced HbA1c declines of up to 2.0% with retatrutide. That control may reduce your risk for heart and kidney complications associated with diabetes.
By lowering liver fat and indicators of liver scarring, retatrutide might stave off the progression of fatty liver disease, which is related to both cardiovascular and kidney health. Retatrutide’s general application in metabolic disease can signify an actual difference for those dealing with more than simply weight problems.
Its impact on hormones and liver health demonstrates it can be used to address issues such as non-alcoholic fatty liver disease, insulin resistance, and metabolic syndrome. As adoption advances, doctors could turn to retatrutide not just for weight loss but for addressing the multiple co-morbidities of obesity.
- Liver Health: Retatrutide can cut liver fat and reduce scarring, which helps stop the progress of liver disease linked to obesity.
- Blood Sugar Control: It improves markers like HbA1c and lowers risks tied to diabetes.
- Hormone Balance: Changes in leptin and adiponectin show a shift toward a healthier metabolism.
- Heart and Kidney Outcomes: By acting on these factors, retatrutide may help prevent long-term heart and kidney disease.
Future Outlook
Retatrutide, a novel tri-receptor agonist-based therapy, is currently going through Phase 3 clinical trials. These trials are examining its efficacy and safety in individuals with obesity and type 2 diabetes. The primary trial intends to recruit approximately 1800 participants and will conclude in February 2026.
Another trial, centered around 405 patients, should conclude in December 2025. Assuming successful outcomes and that regulators approve the drug on time, retatrutide could get into the hands of the public a few years after these trials. The very first launch could be as early as late 2026 or 2027. Approval timelines could vary depending on results, regulatory review velocity, and new safety information.
It’s an obesity epidemic at a global scale. By 2030, projections suggest that approximately 38% of adults will be overweight and 20% obese. That would suggest billions of individuals could be more vulnerable to weight-related diseases, such as diabetes or heart disease. Existing drugs hit one or two hormone pathways, but triple receptor activation by retatrutide might represent a novel path forward.
The obesity treatment market is huge and expanding, and new therapies tend to experience robust demand. Should retatrutide turn out to be safe and effective, it could potentially reshape the future of obesity treatment. This may result in increased alternatives and improved results for those who find it difficult to shed pounds through traditional methods. It could contribute to curbing the increase of obesity-related illnesses, which chop off as many as seven years from the life span.
Research doesn’t end when a drug is launched. This encompasses its long-term effectiveness, potential side effects, and differential impacts across various populations. For instance, outcomes could differ for type 2 diabetics compared to others. Scientists are monitoring how these treatments impact overall health, beyond just weight.
Its long-term data is what helps doctors and patients make informed decisions. It guides health systems on how to deploy new therapies optimally. The future of research into retatrutide and drugs like it is expansive. We hear a lot, for example, about the gut-brain axis and about the role of hormones in appetite and metabolism.
GLP-1 receptor agonists kicked off this trend, but now triple agonists such as retatrutide are being explored beyond obesity. Research might focus on prevention, early intervention, or application to other diseases like heart disease. There is a drive to discover treatments that are simpler, less toxic, or provide additional advantages, like enhanced glycaemic control.
As knowledge expands, more treatments are bound to enter the market, providing new hope to millions.
Conclusion
Retatrutide combines three different hormone targets to help regulate blood glucose and promote weight loss. Its structure allows it to hit multiple systems simultaneously. Trials indicate it could assist adults battling obesity or type 2 diabetes. Side effects pop up but remain generally mild or manageable. The drug may hold promise for heart health and liver issues. Trials continue, so we shall have more information. If you’re anyone who wants to know if this fits their health plan, talk with a doctor. Keep up with new research to see how triple-agonist drugs could transform treatment for diabetes and obesity. Stay updated and consult health professionals for further information.
Frequently Asked Questions
What is retatrutide triple receptor activation?
Retatrutide activates three hormone receptors: GLP-1, GIP, and glucagon. This triple action aids in blood sugar regulation, appetite control, and energy expenditure, providing a promising approach to weight loss and metabolic health.
How does the triple-agonist mechanism work?
Triple-agonist means targeting GLP-1, GIP, and glucagon receptors. This multi-pronged action enhances insulin sensitivity, curbs appetite, and increases energy expenditure, setting it apart from existing mono- or dual-agonists.
What makes retatrutide’s molecular design unique?
Retatrutide is designed to activate three hormone pathways simultaneously. Its design enables selective receptor binding to GLP-1, GIP, and glucagon receptors to optimize metabolic impact balanced with side effects.
What are the clinical implications of retatrutide?
Retatrutide could provide better results for individuals suffering from obesity or type 2 diabetes. Earlier clinical trials have demonstrated impressive weight loss and superior blood sugar control compared to current options.
Is retatrutide safe to use?
Early clinical trials indicate retatrutide is safe and well-tolerated. Typical adverse effects consist of nausea and digestive unease. Long-term safety is being studied in ongoing trials.
Does retatrutide offer benefits beyond weight loss?
Yes. Retatrutide could improve blood sugar control and assuage risks associated with metabolic disorders, such as cardiovascular complications. Studies have yet to validate these benefits.
What is the future outlook for retatrutide?
Retatrutide is still being studied. If future studies continue to support its efficacy and safety, it may emerge as the premier treatment for obesity and type 2 diabetes globally.