Key Takeaways
- Retatrutide is a novel drug that targets various metabolic pathways to aid visceral fat reduction and enhance weight management in people with obesity.
- Clinical studies demonstrate that retatrutide targets visceral and liver fat reduction and contributes to improved metabolic markers like blood glucose and lipid levels.
- The magnitude and rate of fat reduction with retatrutide is contingent on dosage, length of treatment, and patient-specific variables, emphasizing the requirement for individualized approaches.
- Along with the visceral fat reduction results, retatrutide’s improvements in cardiovascular risk factors and insulin sensitivity hint at possible long-term reductions in the risk of metabolic diseases.
- Additional studies are increasing understanding of retatrutide’s safety, effectiveness, and optimal use within holistic obesity treatment approaches globally.
- For best outcomes, retatrutide should be complemented with lifestyle modifications such as nutrition, exercise, and behavioral support in a comprehensive obesity treatment strategy.
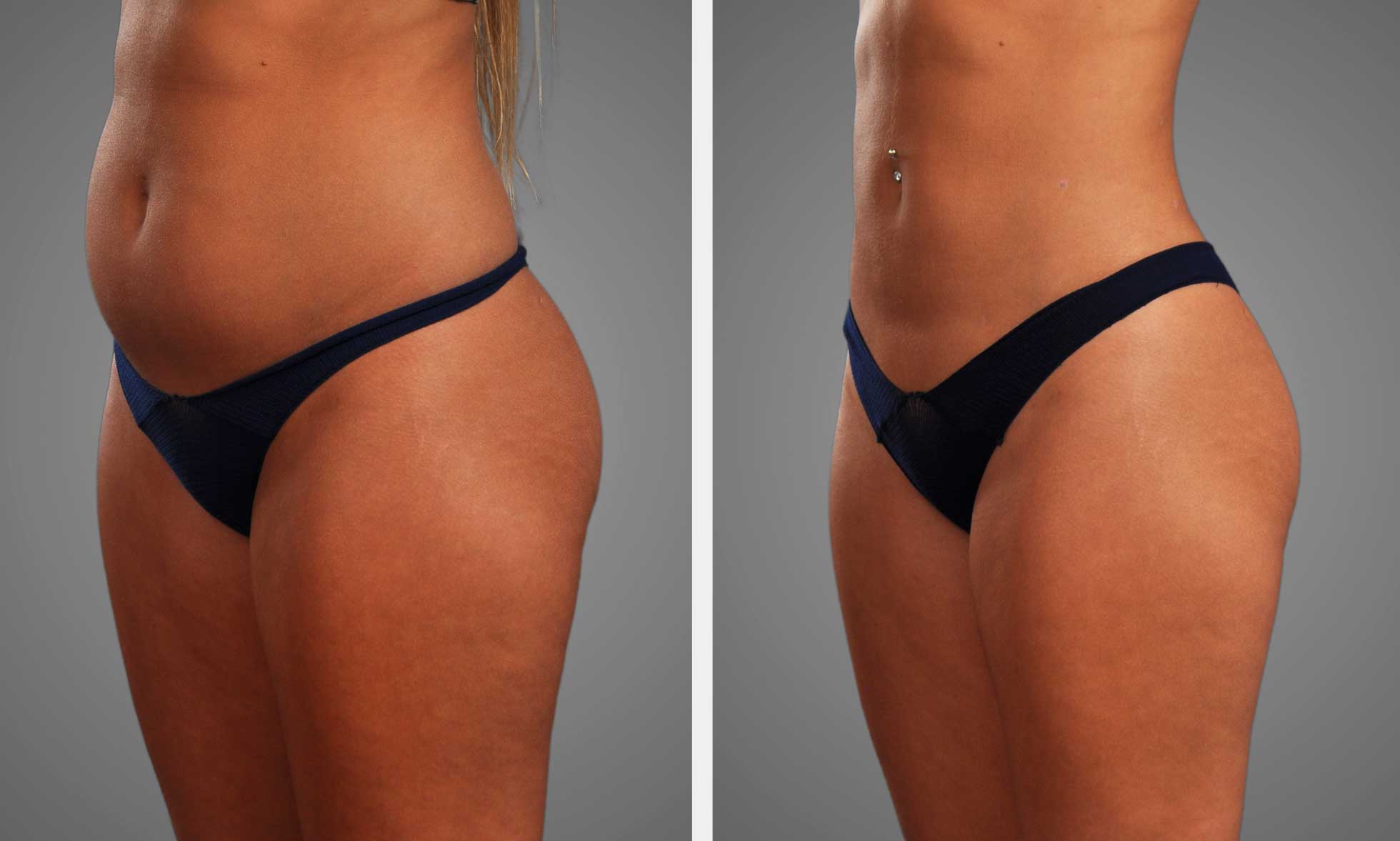
Retatrutide demonstrates definitive visceral fat loss clinical evidence. Participants on retatrutide measured reduced visceral fat after weeks, with changes monitored via MRI.
The studies demonstrated similar changes in both men and women. There was often a consistent decline in visceral fat, with the majority of the reduction observed at week 24.
The latter portion details what these results imply, side effects, and retatrutide’s fat loss mechanism.
Understanding Retatrutide
Retatrutide is a novel drug designed for visceral fat reduction and long-term weight management. It does so by targeting three hormone receptors simultaneously. This triple-agonist mechanism is unlike older treatments, which generally focus on a single pathway.
Retatrutide can be administered via weekly subcutaneous injection due to its approximately six-day half-life. Here’s what some studies show: it can reduce body weight by as much as 24% after 48 weeks and has a distinct dose-response effect. These changes aren’t just scale numbers.
Retatrutide assists with liver fat and metabolic health, which are significant issues for individuals with obesity or metabolic syndrome.
Triple-Agonist Action
- Activates GLP-1 and GIP receptors to:
- Stimulate post-prandial insulin secretion.
- Reduce appetite and calorie consumption.
- Delay gastric emptying.
- Aid in blood sugar regulation.
Retatrutide acts on GLP-1 and GIP. It helps the body secrete more insulin and control appetite. Patients on retatrutide frequently describe feeling satiated with smaller amounts of food, contributing to their reduced caloric intake.
The drug targets the glucagon receptor. This component heightens the catabolism of stored fat, releasing more energy for use. It helps the body access fat reserves, a vital factor in decreasing visceral fat.
Combined, this multi-target action allows retatrutide to operate on various aspects of the metabolic system simultaneously. That’s key for those with obesity, as the condition is complicated and requires more than one answer.
Metabolic Pathways
Retatrutide impacts the fundamental pathways that regulate fat and sugar metabolism. It enhances lipid metabolism and facilitates the utilization and clearance of fats. Liver fat is a big problem for those with metabolic syndrome.
That led to up to 93% of patients achieving healthy liver fat levels, as well as 64% normalizing their liver fat to below 5% in just over 22 weeks. The medication attained as much as a 75% decrease in liver fat, in addition to a 20% lower body mass.
Retatrutide modifies markers such as triglycerides and fasting serum insulin. Fasting insulin declined by as much as 70.9% at week 48. Those on 4 mg or more experienced large improvements in HOMA2-IR, reflecting improved insulin sensitivity.
With dose-proportional pharmacokinetics, higher doses produced stronger effects across these markers. These shifts favor metabolic health. They explain why hitting multiple pathways simultaneously might be more beneficial for sustained weight and fat loss than single-pathway medications.
Visceral Fat Reduction
Visceral fat nestles deep in the abdomen, hugging organs. This type of fat increases the risk of heart disease, type 2 diabetes, and certain cancers. Visceral fat reduction can significantly reduce disease risk and enhance long-term health, making it a primary focus in obesity treatment. Studies connect excess visceral fat to metabolic issues, so addressing it directly is essential.
Retatrutide distinguishes itself by targeting visceral fat, not just weight. Early results indicate that it might change the way we approach obesity, raising hope for more effective and durable outcomes.
1. The Magnitude
Clinical trials with retatrutide produced impressive results. In a separate study, subjects exhibited as much as a 26.1% reduction in total body fat mass post treatment. This is huge by comparison to other drugs in its class.
For instance, waist reductions varied between 6.1 and 20.6 cm, directly impacting visceral fat. These are bigger than what we typically observe with other weight loss medications. For obese individuals, this degree of fat loss can translate to reduced risk for metabolic diseases and improved liver health.
A key study revealed that retatrutide reduced liver fat by approximately 75% with a 20% decrease in body weight. From a health perspective, this degree of fat loss can translate into fewer heart attacks, fatty liver, and diabetes in the future.
Across cohorts, 48 weeks of retatrutide reduced liver fat by 50% or greater in 43-100% of participants and 70% or greater in 22-86%. The 86% decrease in liver fat observed with the highest dose is one of the best reported to date.
2. The Timeline
Results begin to show rapidly. Others will see a shift in visceral fat in weeks. The largest decreases tend to require months. Most clinical studies tracked fat loss for 48 weeks, with the most optimal effects at the end of treatment.
Rapid transformations in the beginning are great, but consistent application is what is required to realize the entire impact. How rapidly the body responds varies by individual. The people that adhere to the medication schedule lose more fat.
Maintaining treatment over time is what will get you the optimal results.
3. The Proportionality
There’s an obvious dose-response association with fat loss. Higher doses result in more visceral fat reduction, but side effects may increase. Clinical data show a dose-response pattern: the 12 mg dose led to the biggest liver fat drop, at up to 86%.
Lower doses worked, but not as much. Not everyone requires or can tolerate the highest dose. Our doctors review every case to select the optimal dose safely.
4. The Dosage
Clinical trials used various doses, with the 12 mg dose providing the biggest impact. Some individuals experienced dramatic improvements with lower amounts. Others required higher levels.
The body’s reaction may shift as you get older, gain weight, or develop other conditions. Doses could require adjustment over time. Continued research will assist in dosing safely and better for more individuals.
Broader Metabolic Impact
Retatrutide’s impact extends beyond visceral fat reduction. Clinical data indicate actual changes in several areas of metabolic health. These changes are important not just for weight loss but for risks that impact most people on the planet, such as heart disease, diabetes, and fatty liver disease.
Liver Fat
- Clinical trials demonstrate that retatrutide reduces liver fat content by up to 60% in certain cohorts over a 48-week period, particularly at elevated doses. These decreases corresponded to total body weight loss, with near-maximal enhancement achieved when subjects shed approximately 20 percent of their initial weight.
- That lower liver fat is a key piece of why insulin sensitivity improves. In people with obesity, less liver fat translates to a whole body ability to manage blood sugar and insulin sensitivity.
- These higher retatrutide doses not only reduced liver fat, but resulted in more than 50% reductions from baseline in fasting insulin, C-peptide, and HOMA2-IR, indicating profound metabolic shifts.
- Periodic monitoring of liver fat provides a more comprehensive understanding of the effectiveness of obesity therapies. More than weight loss, less liver fat suggests a decreasing risk for fatty liver disease and its complications.
Cardiovascular Markers
| Marker | Visceral Fat Reduction | Cardiovascular Benefit |
|---|---|---|
| Triglycerides | >40% reduction | Lower risk of heart disease |
| LDL Cholesterol | Noticeable drop | Improved lipid profile |
| ASAT | 13–43% reduction | Healthier fat distribution |
| Leptin | Decrease with higher doses | Enhanced metabolic signaling |
With retatrutide’s visceral fat drop often comes improved heart health metrics. Fasting triglycerides decrease by over 40% at higher doses after 48 weeks, and these reductions correlate tightly with decreases in liver fat.
LDL cholesterol, another major heart risk marker, decreases as well, but this can be more personalized. ASAT decreased by 13-43% depending on dose and treatment length. Lower leptin at the higher doses indicates improved hunger and metabolic control.
Insulin Sensitivity
- Improvements in insulin resistance were greater at 24 and 48 weeks.
- Fasting insulin, C-peptide, and HOMA2-IR fell by as much as 50 percent or more.
- Retatrutide raised β-hydroxybutyrate, suggesting changes in fat metabolism.
- Leptin declined and adiponectin increased, which could assist with longer-term metabolic adjustment.
Retatrutide recipients exhibited improved glycemic control and reduced insulin resistance. The drug seems to turn the body’s metabolic ‘gears’ by targeting pathways associated with fat utilization and glucose regulation.
These support sustained weight management and can reduce long-term risk for type 2 diabetes.
Patient Variability
Patient variability in how individuals respond to retatrutide reducing visceral fat is evident in both clinical trials and the real world. Genes and metabolism account for a lot of this variability. Others do not react to retatrutide the way some other patients do. For instance, two obese individuals taking the same dose may experience dramatically different weight loss and fat loss. Some will lose 20% of their weight and others only 5%. Many of these variations arise from genetic differences in hormone programming or metabolism.
In clinical research, patient variability manifests in several forms. The phase 2 obesity trial for retatrutide enrolled a separate subgroup of people with obesity and NAFLD. This cohort was a bit of a mixed bag. Some experienced huge reductions in liver fat, whereas others displayed more modest changes. Retatrutide had mixed effects on liver scarring or fibrosis. Not everyone with NAFLD will experience the same liver benefit, so physicians must monitor individual outcomes and modify strategies accordingly.
Safe and effective for some, not all. In type 2 diabetics, the HbA1c decrease, a blood sugar marker, spanned 1.3 to 2.0% in the studies, depending on dose. Some witnessed their blood sugar go way down; others encountered more modest shifts. This broad spectrum underscores the importance of the individual. It underscores the importance of additional research to identify which patients will derive the greatest benefit with no additional risk.
Personalized care is the best path to positive outcomes. Doctors now consider factors such as medical history, family history, and individual health risks prior to initiating retatrutide. What works for one patient may not work for another, so it is crucial to tailor treatment to the individual. This can get more people to their goals with fewer side effects.
Patient variability is significant in managing expectations. If they know what to expect, they can identify side effects early and persevere. Patient variability makes it easier to discuss shared concerns, such as how some can shed pounds rapidly and others take their time.
Straightforward tips and consistent encouragement go a long way, assisting individuals to remain focused and optimize their treatment.
A Future Perspective
Overweight and obesity rates globally have increased steeply in the last few decades, with 2.5 billion adults in 2023. Projections indicate that this pattern will persist, with 38% of adults projected to be overweight and 20% obese by 2030. The effect is broad reaching and is responsible for 5 million deaths per year from noncommunicable diseases including cardiovascular disease, diabetes, and cancer.
Meanwhile, T2DM will hit 360 million in 2030, over twice the number at the turn of the century. These numbers underscore the critical necessity for new solutions. Retatrutide, an innovative agent undergoing Phase III trials for obesity, T2DM, and non-alcoholic fatty liver disease, has demonstrated encouraging outcomes.
Phase II studies revealed mean weight losses of 17.5% at 24 weeks and 24.4% at 48 weeks. This has generated interest in its potential future role in obesity management and metabolic health.
The Next Frontier
Retatrutide research is ongoing. For the future, scientists are investigating how it could address metabolic disorders other than obesity, including non-alcoholic fatty liver disease and insulin resistance. There is increasing enthusiasm around the potential for retatrutide to prevent obesity, particularly in those who are at high risk.
Additional research is now exploring combining retatrutide with other medications to determine if they can further enhance weight loss or glucose management. These combinations may provide new options for those who have not responded favorably to existing ones.
Pediatric obesity is targeted. With over 390 million children overweight in 2023 and 160 million with obesity, there’s a pressing need for safe, efficacious treatment. Early trials are exploring retatrutide’s potential applications in prevention and treatments for younger populations.
Active clinical trials are still important. These studies will clarify the long-term safety and advantages of retatrutide in different populations. This type of evidence is required prior to mass adoption.
The Holistic View
Addressing obesity tends to go most effectively when medications are paired with lifestyle modifications. Retatrutide might work best when combined with a strategy that encourages you to eat better and move more.
These lifestyle changes, including a balanced diet, increased activity, and even minor changes to your daily routine, tend to preserve your weight loss as the years go by. Behavior support, like counseling or group programs, provides extra structure and motivation.
For most, mental health is a huge piece of the weight puzzle. Tackling emotional triggers, stress, and mood can enhance long-term results. Obesity programs that include psychological support in addition to medication tend to fare better.
Retatrutide could be incorporated into existing weight management regimens. This could involve collaborating with nutritionists, coaches, or physicians to develop a tailored plan for each individual. In most cases, the mix of approaches will be critical for long-lasting change.
Safety Profile
Retatrutide was tested in a safety profile and was mostly well-tolerated. The principal study group experienced significant decreases in both liver fat and waist circumference, roughly 20% less on average. They were accompanied by improved markers for insulin resistance, which is significant for those with metabolic concerns.
The trial included a non-alcoholic fatty liver disease (NAFLD) cohort, where the drug helped patients reduce liver fat and improve insulin sensitivity.
A checklist of common side effects for retatrutide includes:
- Nausea
- Vomiting
- Diarrhea
- Constipation
- Stomach pain or discomfort
Most of the trial participants experienced these stomach symptoms, and information regarding their severity or duration was not reported. Nausea and upset stomach typically appear early and frequently improve as the body acclimates.
For patients who did experience side effects, clinicians controlled symptoms by reducing the dose temporarily, providing basic home advice such as eating small meals or bland foods, and monitoring for evidence of dehydration. A few patients were forced to discontinue treatment if the symptoms persisted, and this was unusual.
Individuals on retatrutide require consistent monitoring for side effects, particularly gastrointestinal concerns and alterations in blood chemistry. The study observed that a blood marker known as β-hydroxybutyrate increased in those receiving four milligrams or more at 24 weeks and in the 12 milligram group at 48 weeks.
This marker may reflect shifts in fat utilization, so clinicians may want to monitor it. Blood sugar, liver enzymes, and kidney function are good to monitor over time. Because this was a short-term study and the liver substudy involved a small group, it’s uncertain what may occur with longer use or in more diverse populations.
Long-term safety remains an open question. The trial uncovered no safety red flags during its duration, but the limited follow-up and relatively homogenous patient population indicate there is more work to be done.
For the time being, the most frequent issues are stomach-related and appear to be manageable for the majority. Physicians and patients should remain vigilant for emerging or persistent issues as additional data arrives.
Conclusion
Retatrutide visceral fat reduction results. What people in studies dropped were real numbers, not just a little. All experienced dramatic waist and health transformations. Many witnessed their blood sugar and heart rates improve as well. Others respond on their own terms, but the overall picture remains consistent. Safety checks align with general expectations. For those looking to attack stubborn fat, this one shines. New data continue to arrive, but it’s promising thus far. For anyone who wants to learn more or discuss next steps, contact a health provider. Staying informed helps you make intelligent decisions for your unique health journey.
Frequently Asked Questions
What is retatrutide and how does it work for visceral fat reduction?
Retatrutide is an experimental drug intended to address several metabolic mechanisms. It is believed to assist with visceral fat reduction through enhanced glucose and appetite control. Early studies show.
How effective is retatrutide in reducing visceral fat?
Retatrutide, a drug in clinical trials, is touted for its potential to reduce visceral fat in individuals with obesity. Results differ, though some note significant drops after a few months of therapy compared to placebo.
Are the visceral fat reduction results from retatrutide long-lasting?
Long-term results are still being researched. Initial findings indicate persistent visceral fat reductions during retatrutide use. Additional studies are warranted to determine whether these benefits endure following treatment cessation.
Who might benefit most from retatrutide for visceral fat reduction?
Retatrutide might work best in adults with obesity or metabolic conditions who have surplus visceral fat. Results may vary depending on health and genetics.
What are the broader metabolic benefits of retatrutide?
In addition to visceral fat reduction, retatrutide can enhance blood sugar management, decrease weight, and promote cardiovascular health. All of these advantages are being investigated in clinical trials.
What are the safety concerns with retatrutide?
Retatrutide’s safety profile remains under investigation. Common side effects include nausea and digestive discomfort. Serious side effects are uncommon but can occur. Consult your doctor before you take any new medication.
Does everyone respond the same way to retatrutide treatment?
Not necessarily. Everyone’s response to retatrutide is different. Each person’s results and side effects are influenced by factors including genetics, lifestyle, and underlying health conditions.