Key Takeaways
- Clinical trial evidence finds cryotherapy post-surgery reduces pain and swelling and is most effective as a complement to conventional care to optimize healing and minimize complications. Administer timed cold bursts and pair with prescribed pain meds.
- Cryotherapy lowers pain scores and often reduces additional analgesic use. Continuous flow devices and compression systems generally provide more consistent relief than simple ice packs. Choose a modality based on procedure type, availability, and patient tolerance.
- Cryotherapy effectively decreases edema, bruising and inflammatory markers by promoting vasoconstriction and decreasing nerve conduction velocity, which is extremely helpful after rhinoplasty, facelifts and liposuction. Keep an eye on skin temperature and don’t stay so long that you cause cold injury.
- Cryotherapy after cosmetic surgery evidence points to enhanced scar quality and potentially less hypertrophic scarring by modulating collagen synthesis. Benefits differ by technique and timing. Incorporate cryotherapy into a comprehensive scar-management approach. Consider intralesional or targeted modalities for established problematic scars.
- Established protocols aim at safe skin temperatures and balanced duration and frequency. For example, brief intermittent sessions during the initial post-op week are preferred instead of extended continuous cooling. Modifications are necessary for elderly patients or those with circulatory or sensory impairments. Adhere to device warnings and clinician advice to reduce risks.
- Potential harms include cold injury, nerve irritation, and delayed healing when cooling is excessive or improperly applied. It is important to screen for contraindications like cold intolerance or peripheral vascular disease and discontinue treatment at the initial signs of skin alteration or persistent numbness.
Cryotherapy after cosmetic surgery evidence is the research on cold therapy use to reduce pain, swelling, and bruising after procedures. Trials have mixed results based on treatment, timing, and application method, but a few report modest pain relief and accelerated swelling reduction.
Safety notes skin injury risk with improper use. The meat of the post covers clinical trials, practical protocols, and gaps for future research.
The Scientific Evidence
The scientific evidence for cryotherapy post cosmetic surgery is a mixed, but expanding evidence base. Early work stretches back to the 70s and 80s, such as this 40-year-old study that predates modern RCT standards. Newer trials are better designed but frequently include small samples, short treatment windows, and inconsistent treatment frequency ranging from twice daily to near-continuous.
Cryotherapy has a wide clinical pedigree outside of aesthetics, treating skin cancer, actinic keratosis, and viral warts. It is used alongside physical therapy for musculoskeletal care.
1. Pain Reduction
Controlled trials typically find lower mean pain scores in cryotherapy groups compared to controls. A number of studies demonstrate statistically significant reductions in post-operative pain at early time points, which are hours to the first few days, with mean pain score decreases typically on the order of 20 to 40 percent depending on surgery and device.
Cold slows nerve conduction and local metabolism, which accounts for a lot of the effect. Reduced pain often translates to less need for rescue analgesics. Trials report fewer opioid or nonsteroidal doses in patients receiving regular cold therapy, although the magnitude varies by study.
One limiting factor is that older studies used nonstandard analgesic protocols, so comparisons are uneven. Different devices yield different results. Simple ice packs lower pain acutely, while cold flow devices of either type deliver more consistent, longer duration cooling.
Continuous compression systems provide mechanical support and syndesis may facilitate better pain relief. Peak pain relief usually develops within 24 hours of application and can return once therapy is discontinued, so timing and duration are important for results.
2. Swelling Control
Cryotherapy diminishes postoperative edema by causing microvascular vasoconstriction and reducing capillary leak, which in turn decreases accumulation of tissue fluid. Several studies find less swelling following facial and body surgeries when cold packs are applied early and frequently.
Cold compresses or packs can be beneficial for superficial facial swelling. Even advanced devices that provide uniform temperature and compression frequently have higher volume reductions in comparative studies. Maintaining skin temperature in an effective range is key.
Some studies mention a negative 10 degrees Celsius isotherm at approximately 5 mm from a cold source, demonstrating the impact of depth and temperature profile on outcomes. Regular schedules yield superior edema control compared to intermittent use. The ideal frequency varies between studies.
3. Bruising Minimization
Cold-induced vasoconstriction limits bleeding into tissues and thus reduces bruising. This effect is most pronounced in rhinoplasty, blepharoplasty, and facelifts, where surface ecchymosis is widespread.
They typically find smaller and shorter-lived bruises in their cryotherapy groups compared to standard care. More frequent and longer sessions speed bruise disappearance, but too much cold can damage tissue and must be avoided.
4. Inflammation Modulation
Cold reduces inflammatory mediator release and leukocyte activity, with a small number of studies showing decreased markers post-surgery. Intermittent cryotherapy exhibits more effective anti-inflammatory effects than continuous cooling, which may suppress inflammation even further and risks cold-stress to the tissue.
Lower inflammation typically correlates with swifter wound healing and fewer complications, although this isn’t a universal truth. Some trials have contradictory findings.
5. Scar Quality
Other data indicate that cryotherapy can enhance scar appearance and limit hypertrophic change through collagen synthesis modulation and increased tissue extensibility. Intralesional cryotherapy for scars in aesthetic patients shows promise.
One comparison shows modest improvements in scar appearance compared to dressing alone. Sample sizes are small and follow-up is short. Stronger trials are needed to establish long-term benefit.
Key findings on bruising, inflammation, and scar formation:
- Early cold reduces bruising severity and shortens duration.
- Regular cooling lowers edema and inflammatory markers.
- Cryotherapy may decrease hypertrophic scarring risk.
- Effect size varies by device, timing, and frequency.
- Evidence limited by small samples and old study designs.
Physiological Mechanisms
Post cosmetic surgery cryotherapy works through a number of related physiological mechanisms that all decrease hemorrhaging, swelling, pain and metabolic need at the site of surgery. The subsections below dissect the primary mechanisms: vasoconstriction, nerve conduction alterations, and metabolic suppression, and demonstrate how they synergistically generate clinical advantages.
Vasoconstriction
Cold induces reflex vasoconstriction by activating cutaneous cold receptors and sympathetic pathways that constrict local blood vessels and shut off perfusion to the area. Skin temperature in the therapeutic range of approximately 10°C to 15°C generates significant vessel tone without the elevated risk of frostbite.
The skin cools rapidly initially, then more slowly while deeper tissues cool with a lag. Reduced perfusion limits capillary leakage and interstitial fluid build-up, so edema is lessened and visible swelling drops. Blood flow decrease contributes to reducing postoperative bleeding and hematoma risk since fewer red blood cells and clotting factors arrive to injured microvessels right after surgery.
Different cryotherapy devices produce variable vasoconstriction. Crushed-ice packs cool rapidly but carry a higher skin injury risk, whereas controlled continuous-flow devices maintain target temperatures and more consistent perfusion changes. Localized cryocompression can add pressure to the vasoconstrictive effect, thereby further cutting bleeding and helping bruises remain smaller and fade faster.
Nerve Conduction
Cold decreases nerve conduction velocity by slowing the rate of ion channel kinetics in peripheral nerves. This results in elevated pain thresholds and increased patient comfort following both facial and body treatments. Small myelinated A-delta and unmyelinated C fibers, both important in conducting pain, slow considerably with cooling, while larger A-beta fibers involved in touch are less affected at mild temperatures.
How much slowing occurs depends both on temperature and application time, with colder temperatures and longer applications generating more reduction in conduction and increasing the likelihood of sensory numbness or skin damage. Most effects are temporary: conduction returns as tissue rewarms, which supports a favorable safety profile when protocols limit exposure.
They observe altered pain tolerance and sensory nerve conduction following cryotherapy, but effects differ by method.
Metabolic Rate
Cooling decreases local metabolism and tissue oxygen consumption, thereby diminishing the production of inflammatory mediators and oxidative stress in acutely injured tissues. With a lower metabolic rate, mitochondria produce fewer reactive oxygen species, which help restrict secondary cell damage and improve wound healing dynamics.
Short sessions create modest metabolic drops, and prolonged cooling generates more suppression but may slow reparative cell functions if taken too far. Reduced metabolic demand is particularly salient in regenerative medicine and for graft survival, where temporary metabolic suppression sustains tissue until perfusion is restored.
WBC has been associated with changes in antioxidant capacity, indicating systemic effects. Lowered blood flow, nerve slowing, and metabolic suppression combine to produce the therapeutic profile clinicians rely on post-cosmetic surgery.
Procedure-Specific Benefits
Benefits of Procedure-Specific Cryotherapy after Cosmetic Surgery
Here are procedure-specific effects, evidence comparisons, and practical considerations to orient clinicians and patients on when and how cold therapy can add benefit.
Facial Surgeries
Clear benefits of cryotherapy exist for rhinoplasty by decreasing edema and ecchymosis due to reduced local blood flow and slower nerve conduction velocity, which increases pain threshold and tolerance. Research and clinical experience cite quicker resolution of periorbital ecchymosis with intermittent cold packs for the first 48 to 72 hours.
Facial cooling devices that provide consistent temperatures prevent localized cold injury while maintaining tissue cool. Facelift and eyelid surgery recovery is often managed with controlled cooling. Cold compresses and wearable cooling masks alleviate swelling and pain and can enhance scar appearance by minimizing inflammation and oxidative stress.
Cryotherapy can stimulate endorphin release, providing a natural pain relieving effect that patients commonly describe as mood enhancing in the immediate post-op period. For facial fat grafting and nonsurgical skin tightening, results are procedure-specific. Intermittent surface cooling diminishes immediate postprocedure swelling but if aggressive can blunt graft take.
Contrast protocols or modest localized cooling strike a balance between controlling inflammation and preserving graft survival. For nonsurgical tightening, whole-face cryotherapy versus targeted cold devices show different trade-offs. Whole-face chambers promote systemic antioxidant and immune effects while local cooling focuses on edema control and scar modulation.
Impact on facial nerve function appears minimal when protocols avoid extreme or prolonged local cooling. Small drops in nerve conduction velocity account for some analgesia, but careful temperature control prevents neuropraxia and supports better cosmetic outcomes when applied per guidelines.
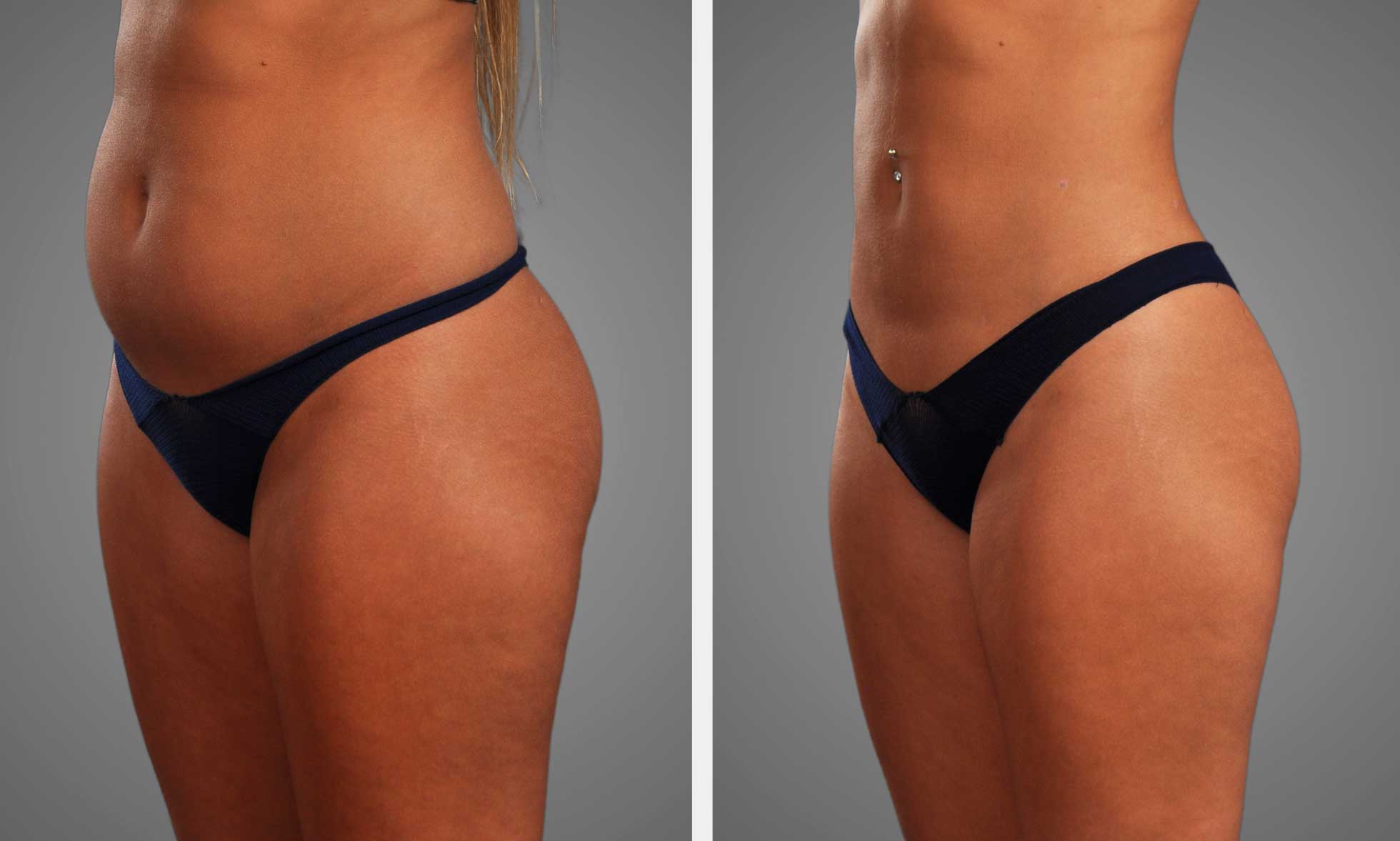
Body Contouring
Post-liposuction and -gluteal lipoinjection, cryotherapy diminishes pain and swelling through decreasing tissue metabolism and inflammation, with many centers noting earlier time to comfortable ambulation. Cryolipolysis and other body-directed cold techniques serve a different role: they target adipocytes for fat reduction rather than immediate postoperative inflammation.
There is evidence behind modest contour improvement with cryolipolysis, but it’s no replacement for surgical aspiration. Surface cooling is usually safer and more convenient to apply over large areas than invasive internal cooling, which can endanger deeper tissue ischemia. Method comparison shows that external devices enable continuous skin temperature monitoring and are less prone to induce cold injury when employed with appropriate padding and timing.
Monitoring skin temperature is especially crucial in large-area treatments. Extreme cold activates thermoreceptors and hypothalamic reactions. Uncontrolled decreases lead to skin tissue injury, delayed healing, or diminished enzymatic activity. Such protocols prevent localized freezing and continue to use intermittent schedules to preserve tissue.
Breast Procedures
Breast implants or augmentation or removal: Cryotherapy reduces pain and swelling and accelerates recovery when utilized as cold packs or cold flow devices. Devices that circulate cooled liquid under a wrap hold constant pressure and temperature, which increases comfort and reduces the risk of hematoma in certain reports.
Protocols for breasts often mirror those for other areas but require lower intensity and shorter cycles to prevent tissue ischemia around implants or flaps. Safe parameters prevent delayed healing while retaining anti-inflammatory and analgesic benefits.
Clinicians must balance cryotherapy’s immune and antioxidant effects with the need to preserve perfusion and tissue repair.
Application Protocols
Application protocols for cryotherapy after cosmetic surgery. Below are standard protocol items numbered and then followed by specific discussion of temperature, duration, and frequency as they pertain to various procedures and patients.
- Standard parameters: target skin temperature, device type, session length, and frequency. Try for a skin temperature that’s low enough to reduce inflammation and pain, but not so low as to cause frostbite or cold panniculitis. Employ controlled appliances wherever feasible. Ice packs are permissible with diligent observation. Common devices include everything from basic gel packs to computer-aided local cryotherapy and whole body cryochambers.
- Temperature guidelines with buffers. A lot of investigative studies have tissue exposures on the far end of the scale, for example, whole-body cryotherapy at minus 110 degrees Celsius to minus 140 degrees Celsius. Clinical postoperative applications concentrate on skin temperatures instead of air temperature. Target skin surface ranges prevent tissue damage.
- How long a session lasts and when. Short, controlled exposures tend to be advantageous. Some protocols employ 3-minute bursts while some report multi-hour application. Match time to device, site and surgical trauma.
- How often and when should an app report its data. In the first 72 hours, several brief sessions typically beat one long session for a lot of outcomes. Some research administers cooling 2 to 3 times per day, or even continuous 2 to 6 hour blocks. Select your own based on tolerance and wound factors.
- Mixtures and additives. Add compression, light activity or physio to your cryotherapy and they will help swelling and mobility. Record experiences with pain medication and bandages.
- Patient- and procedure-specific customization. Modify for age, vascular disease, thin or fragile skin, and technique which liposuction or small excisions. Lower the dose and length for higher-risk individuals.
Temperature
Target skin surface temperature for safe, effective cryotherapy usually sits above the threshold for frostbite and cold panniculitis. Shoot for a monitored range, not absolute air temperature. Research with extreme WBC low air temperatures from minus 110 degrees Celsius to minus 140 degrees Celsius is not the same as local cooling.
With devices varying, even a minus 120 degrees Celsius chamber can still produce safe skin temperatures with regulated exposure. Ice packs generate uneven skin drops, while controlled, computer-assisted systems more effectively maintain skin in the targeted range. Colder skin temperatures provide better short-term pain relief and less inflammation, but if skin falls too low, the risk of tissue damage increases. Controlled protocols and skin thermometers are essential.
Duration
Ideal single-session cooling remains controversial, while lab and clinical evidence favors very short targeted exposures on the order of 3 minutes for maximal neurovascular effect with minimal injury risk. Other clinical protocols report longer times: 30-minute pack applications or even overnight and multi-hour (2 to 6 hours) sessions.
While extended uninterrupted cooling can minimize edema in certain surgeries, it raises the likelihood of cold injury. Shorter, intermittent application protocols can help maintain this benefit while minimizing damage. For deeper surgeries such as large flaps or liposuction, longer or serial treatments might assist but need more careful observation.
Frequency
In the immediate postoperative week, most protocols employ several daily treatments, typically 2 to 3 times per day, with increased frequency during the first 48 to 72 hours. Single long freezes show mixed results against multiple shorter sessions.
Multiple sessions often control swelling and pain better. Adjust frequency by procedure: minor excisions may need only one daily session, while major reconstruction benefits from closer, individualized plans. Monitor patient reaction and adjust to prevent hyperhypothermia.
Potential Risks
Cryotherapy after cosmetic surgery may lessen pain and swelling but it poses several potential harms that deserve serious consideration prior to, during and after use. These risks range from typical, anticipated side effects to uncommon but significant complications. Clinicians and patients must balance these results with anticipated advantages and watch carefully for issues.
Common adverse effects include transient pain, swelling, blistering, mild erythema, and temporary numbness. Less common but notable risks include cold panniculitis, delayed wound healing, superficial scarring, hypopigmentation, and hyperpigmentation. Rare but serious risks include frostbite, tissue necrosis, permanent nerve injury, secondary infection, and deep tissue damage in vascularly impaired areas.
Procedure-specific risks include recurrence of treated lesions, which ranges from 6 to 34 percent. Variable cure rates for lesions range from 39 to 84 percent at three months, and there is incomplete tumor control for some skin cancers. Device-related risks involve thermal injury from improper device settings, damage to surrounding healthy tissue, and inconsistent cooling with simple ice packs.
Skin Damage
Frostbite, cold panniculitis, and thermal injury can happen if cooling is too aggressive, left on too long, or applied without adequate skin protection. Frostbite coagulates tissue and forms ice crystals and can cause necrosis if not arrested promptly. Cold panniculitis is characterized by tender, inflamed nodules and can last for weeks.

Conventional ice packs have uneven cooling and a higher danger of mild freeze injury if left in direct contact. Professional level cryotherapy systems utilize controlled temperatures and circulation to decrease that risk, but abuse or malfunction still results in surface burns and scarring. Skin surface temperature should be monitored with infrared or contact thermometers during treatment to prevent dangerous drops.
Early indicators that call for immediate discontinuation are intensifying stabbing pain, hard white patches, tight blisters, skin color loss, and sensory changes.
Nerve Issues
Too much cold can rupture peripheral nerves, either by direct cold axonal injury or ischemia due to vasoconstriction. Damage can be transient, known as neurapraxia, or permanent, referred to as axon loss, depending on the intensity and duration of exposure.
Signs are constant numbness, pins and needles, and weakness or ‘frozen arm’ sensation. These signs can show up while undergoing treatment or days afterward.
Device choice changes danger. Continuous-flow systems with regulated pump rates exhibit fewer nerve complications than unregulated ice application. There is a risk of focal nerve palsy if static ice is applied for too long. Select settings—temperature, time, and contact pressure of interface—adapted to anatomy to minimize nerve damage.
Delayed Healing
Cold decreases local blood flow and metabolic rate. Excessive cold can delay wound healing by impairing perfusion and inflammatory response. This effect is potentiated in patients with diabetes, peripheral vascular disease, a smoking history, advanced age, or use of vasoconstrictive drugs.
Comparative studies indicate that certain patients recover equally well with short, localized cryotherapy, whereas long or repeat sessions are associated with delayed healing and increased infection. Balance pain and swelling control against possible delays. Customize protocols to the patient’s risk profile, check wounds closely, and modify protocols for high risk patients.
Beyond The Hype
The history of cryotherapy dates back to ancient Greece, Persia, and Rome, where snow, ice-and-water slushes, and cold water were used to treat various ailments to today’s localized and whole-body systems. Modern promises for quicker healing, less inflammation, and immune enhancements post-plastic surgery tend to get ahead of the data.
Here are targeted, actionable evaluations of where cryotherapy can assist, where the hype outruns the evidence, and what clinicians and patients should consider when integrating it into post-operative care.
Patient Factors
Patient age, skin condition and baseline immune response shape both efficacy and safety. Older skin can have slower blood flow and a thinner dermis, altering how the cold impacts tissue. A previous history of delayed wound healing, connective tissue disease or immunosuppression can diminish benefits or increase risk.
Tolerance matters: cryotherapy can be intense and uncomfortable, particularly for first-time users, which influences adherence.
- Contraindications for cryotherapy:
- Raynaud’s or severe peripheral vascular disease.
- Cold urticaria or serious cryoglobulinemia.
- Open wounds not cleared for cold exposure.
- Diabetic neuropathy, uncontrolled.
- Extreme heart diseases in which stress response is a danger.
- Ongoing infection at treatment location.
Different skin types and comorbidities alter appropriateness. Darker skin per se is not a contraindication, but scars and pigment alterations may respond differently to cold. Patients who tend to keloids require custom plans.
Rheumatoid arthritis patients could experience pain and mobility advantages according to other research. However, mapping those gains onto surgical wound healing is not straightforward. Individualized plans matter: set clear goals, pick timing and dose, monitor tolerance, and adjust based on healing and symptoms.
Device Differences
| Device type | Typical use | Reported effectiveness | Safety notes |
|---|---|---|---|
| Localized cold packs/compression | Small areas, low cost | Modest pain/swelling reduction | Low risk if used properly |
| Cold‑air/cryospray units | Targeted tissue cooling | Variable; operator dependent | Risk of frostbite if misused |
| Whole‑body cryo chambers | systemic exposure | Claims of immune/antioxidant effects | Intense; low adverse event rate overall |
More sophisticated machines incorporate exact temperature control, skin sensors in real-time, programmable cycles, and built-in compression to minimize risk of frost and enhance reproducibility. Price is all over the map. Minimum packs cost very little, while medical-grade systems are costly and need training.
Convenience and clinic flow count. A well-selected device enhances compliance and probably results.
Research Gaps
Protocols vary: temperature, exposure time, frequency, and timing relative to surgery are inconsistent across studies. There’s little evidence for most cosmetic procedures, with most trials concentrating on pain or movement in non-operative conditions.
Certain studies demonstrate decreased pain and better mobility in rheumatoid arthritis, and others find little advantage. Pivotal issues about dose response, long-term influence on scar formation, and immune or antioxidant mechanisms persist.
Serious side effects are uncommon, with approximately 16 recorded adverse events across millions of sessions, but uncommon doesn’t stand in for rigorous trials. We need more randomized, standardized clinical trials to define role, timing, and patient selection.
Conclusion
The present evidence demonstrates minimal and equivocal advantages for cryotherapy post-cosmetic surgery. Some studies indicate a short-term reduction in pain and less swelling. Blood flow decreases and inflammation markers decrease immediately after cold application. These effects vary by surgery type, timing, and how cold is applied.
Select easy things that complement the procedure. For small spots, short ice packs do the trick. For bigger locations, managed gadgets provide stable temperatures and less skin hazard. Look out for numb skin, discoloration, or persistent pain. If you have circulation or nerve issues, you are at increased risk.
Use cryotherapy as one tool in a recovery plan, not a panacea. Discuss with the surgeon timing and dose. I suggest that you attempt a brief test session and monitor pain, swelling, and skin symptoms. If you need assistance weighing options or making a plan, talk to a clinician or bring your notes to your next visit.
Frequently Asked Questions
Is there strong evidence that cryotherapy helps recovery after cosmetic surgery?
Available evidence is very scant and conflicting. Limited studies indicate advantages in pain and edema. There is a shortage of robust randomized controlled trials. Employ cryotherapy as an adjunct, not a surrogate for standard postoperative care.
How does cryotherapy reduce pain and swelling after surgery?
Cold decreases local blood flow and nerve transmission. This reduces inflammation, swelling, and pain perception. Effects are typically transient and are dependent on application time and temperature.
Which cosmetic procedures benefit most from cryotherapy?
Surgeries with soft-tissue swelling, like rhinoplasty, eyelid surgery, and liposuction, could get the biggest benefit. Depending on the procedure and quality of the study, the evidence is mixed.
What is a safe cryotherapy application protocol after surgery?
Short sessions of 10 to 20 minutes multiple times a day are typically advised. Shield skin with a barrier and adhere to your surgeon’s particular directions. Avoid ice for a long duration or directly.
What are the main risks of cryotherapy after surgery?
Skin frostbite, more bruising if misapplied, and disruption to wound healing are potential risks. People with circulation or nerve disorders should skip it or check with their surgeon.
Can cryotherapy prevent long-term scarring or improve final aesthetic outcomes?
There is no reliable evidence that cryotherapy alters long-term scarring or the final cosmetic result. It primarily provides brief symptom relief while you’re healing.
Should I ask my surgeon before using cryotherapy?
Yes. Your surgeon knows your surgery, incision, and medical history. They can provide personalized advice and safe timing for cryotherapy use.