Key Takeaways
- Retatrutide mimics GLP-1, GIP and glucagon to hit multiple metabolic pathways, which could enhance weight loss relative to single- or dual-hormone medicines. Think of this option if more weight loss is the clinical aim.
- Glucagon receptor activation raises resting energy expenditure and helps drive fat burning, fueling even more efficient body fat loss and potential sustained weight management.
- GIP and GLP-1 activity together promote improved glucose regulation, appetite control, and delayed gastric emptying that can enhance metabolic health while combating calorie absorption.
- With clinical trials reporting significant findings, some showing nearly 29% average weight loss over 16 months and decreased knee pain, retatrutide appears promising for obesity and weight-related joint symptoms. Monitor metabolic markers and functional outcomes throughout therapy.
- While it’s generally well tolerated, common side effects are nausea and GI symptoms with more rare risks such as gallbladder and pancreatic issues. Therefore, monitor your patients and titrate doses slowly to improve tolerability.
- Retatrutide is administered as a weekly subcutaneous injection and is not yet broadly authorized. Track regulatory news, confirm candidate eligibility, and continue lifestyle interventions to support benefits.
Its role in extreme weight reduction is as a new peptide therapy that reduces body weight by influencing appetite and metabolic processes. In clinical trials, it can lead to a few percentage points of weight loss every month, often in combination with diet and activity modifications.
Side effects include nausea and mild GI symptoms, and medical supervision is advised for high-risk cases. Additional sections discuss mechanisms, trial data, safety profiles, and practical considerations for patients and clinicians.
Triple-Hormone Action
Retatrutide mimics three key hormones that regulate hunger and metabolism: GLP-1, GIP, and glucagon. This triple action is different from most existing therapies, which typically focus on one or two pathways. Activating all three receptors at once, retatrutide seeks to enhance weight loss through several simultaneous metabolic actions, including reduced hunger, increased energy expenditure, and more effective glucose metabolism.
1. Glucagon Receptor
In its activation of the glucagon receptor, it increases energy expenditure and encourages lipolysis even at rest. In other words, your body can torch more fat without additional activity, which is great news for couch potatoes and anyone who has a hard time working out more.
Glucagon-driven metabolic rate increases differentiate retatrutide from appetite-lowering-only drugs. By targeting this receptor, it can thus more efficiently reduce body fat and can potentially maintain weight loss for the long haul by keeping resting energy demand elevated over time.
2. GIP Receptor
It is at least a partial agonist at the GIP receptor, which means it stimulates this receptor’s activity as well, not simply inhibits it as an antagonist would. Improved insulin action means the body processes food more efficiently and may reduce the risk of further metabolic disease.
GIP action can enhance satiety signals, reducing food binging in reaction to meals. Along with differences in glucose regulation, GIP participation promotes more widespread metabolic well-being outside of mere weight loss.
3. GLP-1 Receptor
GLP-1 receptor activation is an established mechanism for decreasing appetite and food consumption. It decelerates gastric emptying so people feel full longer after meals, which reduces caloric consumption.
Several of the current weight loss drugs utilize GLP-1 pathways, and retatrutide preserves that advantage while introducing two additional mechanisms. GLP-1 remains a significant factor in calorie reduction with retatrutide.
4. Synergistic Effect
Simultaneous activation of GLP-1, GIP, and glucagon creates synergy: appetite falls, energy use rises, and glucose handling improves at once. That triple action might lead to more rapid and greater weight loss than single- or dual-target drugs.
Clinical results support this: retatrutide produced up to 24.2% weight loss at 48 weeks in a phase 2 trial, and dramatic liver fat declines—mean relative changes at 24 weeks were −42.9% (1 mg), −57.0% (4 mg), −81.4% (8 mg), and −82.4% (12 mg), with normal liver fat achieved in up to 86% (12 mg) of participants.
Non-HDL cholesterol, triglycerides, hsCRP, and fasting insulin were also observed, with up to 70.9% reduction at 48 weeks. Benefits must be weighed against safety: discontinuation due to adverse events occurred in 12.2% and 18.2% for 9 mg and 12 mg doses versus 4.0% for placebo.
A comparison table assists clinicians and patients in contrasting retatrutide with single- and dual-agonists for efficacy and risk.
Clinical Results
Clinical trials continue to show that retatrutide generates dramatic, clinically significant weight loss for individuals with obesity. Across several studies and time points, these trials show rapid and sustained weight loss, metabolic marker improvements, and downstream benefits like less joint pain. The subsections below detail weight change, metabolic impacts, and safety observations from the trials.
Weight Reduction
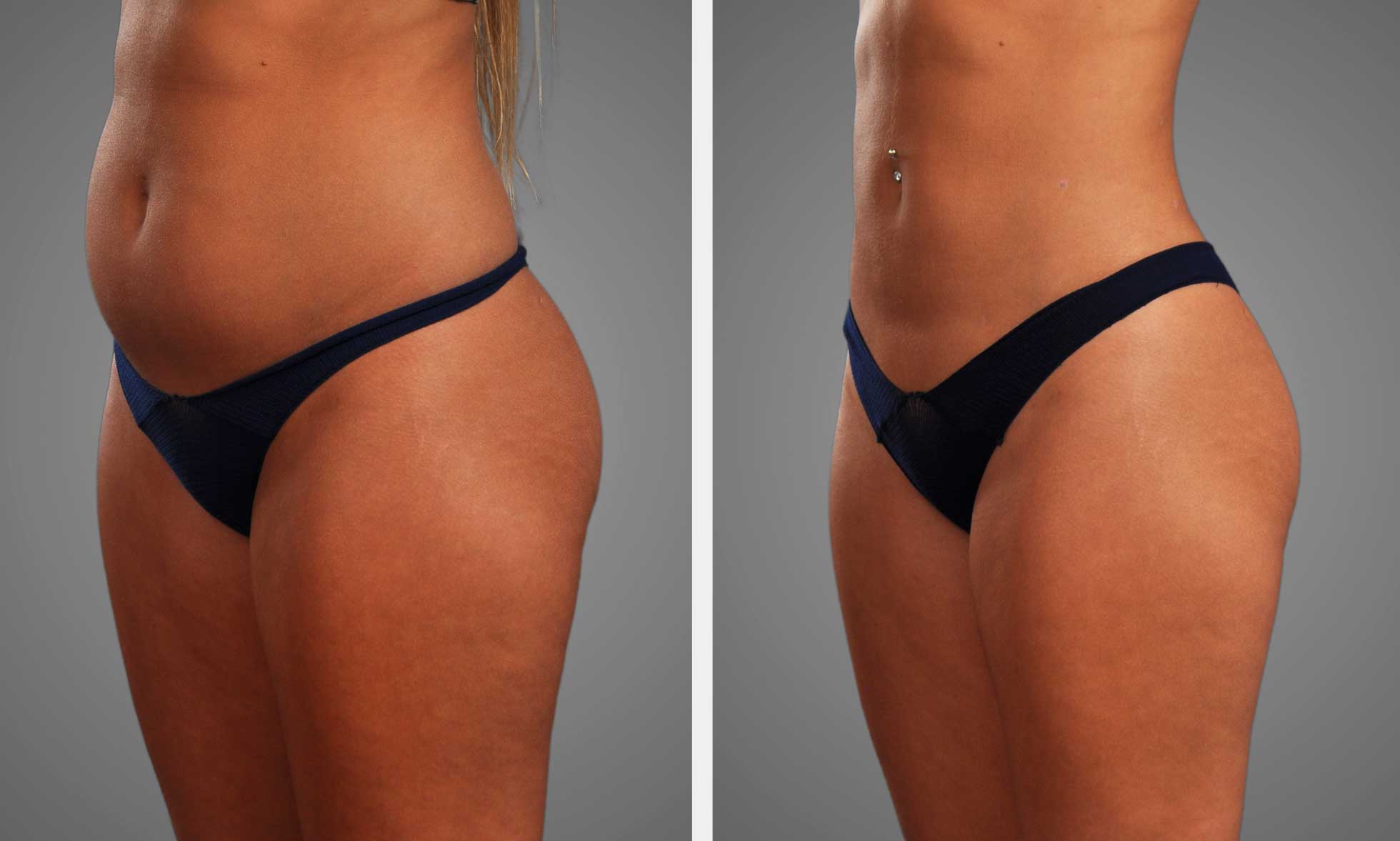
Retatrutide delivered almost 29% mean weight loss at 16 months in a landmark trial, with subjects shedding an average of 32 kg (71 pounds). That level of loss is much more than most approved non-surgical drugs manage. Many GLP-1s, for instance, deliver only double-digit percent loss at best over similar timeframes. Weight losses of this magnitude are rare outside of bariatric surgery, making these clinical results impressive for a pharmacologic treatment.
- Average weight loss is approximately 29% at 16 months, with a mean of about 32 kg.
- Phase 2 comparator: up to 24.2 percent weight reduction at 48 weeks in other cohorts.
- Dose response: One trial reported a bodyweight reduction of up to 16.94% at 36 weeks for the highest dose compared to 3.00% with placebo.
- Time course: sustained and greater losses occur at later time points, consistent with stepped improvements through week 48.
These results imply a dose-dependent impact and a directionality where bigger gains build up with extended exposure. For instance, participants who experienced modest weight loss at 12 weeks saw much more predominant loss at 36 to 48 weeks.
Metabolic Health
Retatrutide’s trials showed benefits beyond weight. Knee pain lessened roughly 76% in another study, resulting in improved movement and a more effortless participation in daily activity. Improvements in joint pain can mean increased activity and increased quality of life.
Metabolic markers improved. Studies saw decreased fasting serum insulin concentrations by as much as 70.9% across week 48 versus baseline, and measures of insulin resistance improved at 24 and 48 weeks, with more change at week 48. Β‑Hydroxybutyrate increased at 24 and 48 weeks, signaling changes in substrate utilization.
Liver outcomes were favorable: trials found reductions in liver fat content, with near-maximal liver fat drop of 75% coincident with 20% bodyweight loss. Exploratory analyses showed numerous participants achieved 50% or greater liver fat reduction or liver fat less than 5%. Fewer participants had MRIs at week 48 versus week 24, which impacts some of the imaging-based endpoints.
Follow glucose, insulin, liver fat, and ketone markers during treatment to measure these metabolic improvements.
Safety Profile
Common side effects are shared with similar agents. Nausea, vomiting, and other stomach issues occur at appreciable rates. Certain reports mention constipation, gallbladder events, and infrequent pancreatic disease.
Closely monitor patients for adverse events and offer definitive instructions to combat nausea and gastrointestinal side effects.
Checklist to monitor:
- Symptoms: persistent nausea, vomiting, abdominal pain. Note onset and severity.
- Bowel changes include constipation frequency, stool form, and the need for laxatives.
- Gallbladder: Right upper quadrant pain, jaundice, or abnormal liver tests.
- Pancreatitis signs: severe persistent abdominal pain, elevated pancreatic enzymes.
- Labs/imaging: Fasting insulin, glucose, liver fat assessment, ketone levels, and periodic imaging where indicated.
Ideal Candidates
Retatrutide is most appropriate for adults who have obesity or have overweight with weight-associated co-morbidities. Clinically, ideal candidates tend to have a BMI of 30 kg/m2 or 27 kg/m2 with one or more comorbidities like type 2 diabetes, hypertension, or dyslipidemia. This category encompasses individuals whose weight adds to daily restrictions, such as those with diminished mobility or diet-resistant, exercise-resistant, and first-line treatment-resistant metabolic risk.
Include practical examples: a 45-year-old with a BMI of 32 and poorly controlled blood pressure despite two medications, or a 38-year-old with a BMI of 28 and new-onset type 2 diabetes who has struggled to lose weight with lifestyle change.
Patients with weight-related joint pain, such as knee osteoarthritis, may receive the highest functional reward. A weight loss of 10 to 15 percent can reduce knee load and improve pain and walking capacity. A patient in their 50s with a BMI of 34 and symptomatic knee osteoarthritis who can’t take long walks or do low-impact exercise might be a good candidate, assuming the other safety checks are clear.
Because of its promise for fast, significant weight loss, retatrutide is an option when the objective is to reduce mechanical stress and postpone or prevent joint surgery.
Exclude individuals with known contraindications to hormone or incretin-like therapies. Individuals with a personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia type 2 (MEN-2) should not be administered these agents. Type 1 DM patients are generally not the best candidates for retatrutide-like regimens.
Exclude those with recent major cardiac events. A history of heart attack, stroke, hospitalization for congestive heart failure, or unstable angina within 90 days prior to screening is commonly used to screen out higher-risk subjects.
Evaluate patient history and comorbidities before starting treatment with careful attention to recent weight change, medication use, and pancreatitis risk. Many trials exclude people who had a change in body weight greater than 5 kg, which is about 11 pounds, within 90 days or who used other weight loss medications, including over-the-counter agents, in that window.
A history of pancreatitis or current pancreatic disease warrants caution. Patients planning bariatric or other surgical weight loss procedures should defer pharmacologic therapy until plans are clear, as surgery may alter risk and benefit.
Pre-treatment assessment should include a cardiac risk review, thyroid family history, glycemic status, recent weight trajectory, and current medications to ensure safe, evidence-based use.
Treatment Protocol
Retatrutide treatment is administered as a regimented, monitored course. The full protocol reported in trials spans 52 weeks: a 6-week screening, 48 weeks of active treatment, and a 4-week safety follow-up. Dosing cohorts were 1, 4, 8, and 12 mg. Most of the patients enrolled in these trials would have had biopsy-confirmed NASH.
Medication should be refrigerated at 2°C–8°C (36°F–46°F). Maintaining the weekly regimen is key to obtaining results observed in trials, which reported liver fat reductions ranging from 51.3% to 86.0% at 48 weeks and weight loss up to 24.2% at 48 weeks.
- Weekly subcutaneous injections.
- Screening, 48-week treatment, 4-week follow-up.
- Dose ranges include 1 mg, 4 mg, 8 mg, and 12 mg.
- Refrigeration 2°C–8°C, proper handling.
- Titration and dose adjustments to limit side effects.
- Routine monitoring: liver fat, insulin, weight, waist.
- Patient reporting and documentation of dosing and symptoms.
Administration
Retatrutide is given once weekly by subcutaneous injection. Educate patients on proper angle, pinching, and single use syringes or pens. Rotate injection sites — abdomen, thigh, upper arm — to minimize local irritation and lipodystrophy.
Weekly dosing is more convenient than daily drugs and could improve compliance among overburdened working adults. Have patients record every dose in a log or app — logs assist clinicians in identifying missed doses or patterns connected to side effects.
Dosage
Dosage is as per treating physician depending on clinical status and response. Begin low and titrate to minimize nausea and other side effects. This makes it more tolerable.
Dose adjustments are typical in the first 12 weeks and can persist as necessary through 48 weeks. The table below shows sample adjustments employed in trials and clinician notes for personal requirements.
| Starting dose | Typical titration steps | Notes for individual patients |
|---|---|---|
| 1 mg cohort | Increase to 4 mg over 4–8 weeks if tolerated | For sensitive patients, stay longer at lower dose |
| 4 mg cohort | Step to 8 mg over 4 weeks if side effects minimal | Adjust down if GI effects severe |
| 8 mg cohort | Move to 12 mg with close monitoring | Consider liver enzymes and glycemic response |
| 12 mg cohort | Maintain or reduce based on safety labs | Pregnant or renal-impaired patients need special review |
Side Effects
Nausea and other GI symptoms are the most common. Trials reported nausea in approximately 27% and diarrhea in 13% of patients. Less frequent but severe risks encompass gallbladder disease and pancreatitis.
Watch out for symptoms such as intense abdominal pain or jaundice. Side effects checklist for patients: Nausea, vomiting, abdominal pain, fever, change in stool. Advise prompt report of serious or protracted symptoms.
Periodic labs and imaging kept us on track by connecting symptom fluctuations to decreases in liver fat and metabolic shifts. In fact, our studies revealed fasting insulin declines as high as 70.9% and a significant correlation between liver fat loss and both body weight and waist circumference reductions.
A Metabolic Reset
Retatrutide works via multi-hormone signaling to rewire fundamental metabolic processes. It acts on receptors that mimic gut hormones that regulate hunger, insulin secretion and lipolysis. By activating several pathways simultaneously, retatrutide is able to reduce food consumption and increase energy expenditure.
Clinical trials note massive weight losses, sometimes as high as around 24% of initial body weight within 48 weeks, and that amount of loss tends to result in widespread metabolic shifts, not just localized weight loss.
Hormone shifts can increase adiponectin, a protein that aids cells in metabolizing glucose and fat more effectively. Research indicates that retatrutide enhances adiponectin, which connects to enhanced glucose regulation and lipid metabolism.
Along with improved adiponectin typically comes less liver fat. A trial demonstrated an 86% relative reduction in liver fat 48 weeks post-treatment. For individuals with MASLD or obesity-related fatty liver, that level of liver fat reduction can decrease inflammation and alleviate short-term liver strain.
Insulin sensitivity improves as weight and liver fat decline. Measures like HOMA2-IR fell, in some reports as much as 54.5%, providing evidence of clearer insulin signaling and decreased fasting insulin demand. Reduced insulin resistance typically reduces both fasting triglycerides and serum C-peptide levels, relieving pancreatic beta cell stress and potentially decreasing diabetes risk down the line.
Those shifts aren’t just biochemical; they alter daily appetite and energy balance, so many patients experience a longer-term decrease in the urge to overeat and less energy crashes.
Something like a metabolic reset ain’t the same for all. The concept of a return to ‘normal’ metabolism is helpful but reductive. Genetics, previous weight history, and lifestyle determine how near someone gets.
Some will require continued medication, others might sustain improvements through lifestyle modification alone. Evidence points to the reset as a process. Initial drug-driven change creates a window where diet, activity, and sleep habits can more easily take hold.
To maintain gains you need to intervene again. Actionable guidance includes incremental fitness regimens that increase muscle and resting metabolic rate, preferential protein and fiber diet plans to blunt appetite, and routine metabolic checkups including liver fat scans, HbA1c, and lipid panels.
With metabolic diseases on the rise worldwide, these actions are timely. Combining retatrutide with structured lifestyle work provides a credible route to lasting change.
Future Outlook
Retatrutide might redefine what’s possible with pharmacological weight loss, providing bigger, faster weight loss than existing alternatives. Clinical trials have documented significant weight loss as well as improved glycemic control, indicating that this triple incretin receptor agonist might redefine what clinicians anticipate from pharmaceutical treatment. This would pivot treatment goals from modest weight loss toward results that compete with surgery for certain individuals.
For those with severe obesity or metabolic disease, a medication that can induce deep weight losses over time may decrease surgical interventions, reduce diabetes rates and associated healthcare costs.
Regulatory timing will determine how quickly that standard can gain a foothold. Approval is improbable prior to late 2026, and deferrals into 2027 or 2028 continue to be feasible. Active trials with one completing in February 2026 will contribute to regulatory reviews and timelines.
Even with a good verdict, the real-world availability is not going to be immediate. According to their estimates, it will take a year or two for new drugs to become widely available as they scale up manufacturing, negotiate prices, and payers make decisions. Clinicians and health systems should anticipate a gradual rollout across countries and care settings.
Comparison studies will come soon after as clinicians and payers want to see how retatrutide compares to today’s leaders. Anticipate side-by-side trials and real-world studies comparing efficacy, safety, durability of weight loss, blood sugar, and cardiovascular risk.
For instance, comparing retatrutide to GLP-1 or dual agonists and testing its role in conjunction with or without lifestyle programs like the Mediterranean diet, which studies associate with improved outcomes in metabolic diseases. Combination approaches, such as drug and diet or weight-management programs, will be tested to discover best practices for long-term adherence and health gains.
Keeping an eye on regulatory updates and trial data will be key for clinicians, payers, and patients. Follow key primary and secondary endpoints from late-stage trials for potential indicators on safety, adverse events, and durability of weight loss.
See if there are any subgroup analyses demonstrating which patients benefit the most—severe obesity, type 2 diabetes, or certain metabolic profiles. Stay tuned for advice on length of treatment, tapering, and coupling with behavioral treatment.
Health systems ought to build pathways for access that take into account cost, equity, and follow-up, because the global burden of obesity will continue to climb through 2030 and beyond.
Conclusion
Retatrutide holds obvious potential for extreme weight loss. By Geoff Watts Retatrutide’s role for extreme weight loss trials document massive, sustained reductions in weight and fat. The drug targets three hormone routes to suppress hunger and increase calorie burning. Patients with critical obesity and risk factors benefit the most. The short-term side effects are generally minor and arrive early. Longer trials will demonstrate how safe it remains and how durable results are.
For clinicians, retatrutide introduces a strong adjunct to diet and activity. For patients, it provides a genuine opportunity to shift health markers quickly. For scientists, it opens new avenues in metabolic medicine. If you want to learn more, read the full trial reports and discuss with a specialist whether this option suits your goals.
Frequently Asked Questions
What is retatrutide and how does it work for extreme weight reduction?
Retatrutide is an experimental injectable drug that targets three hormones: GIP, GLP-1, and glucagon. It suppresses appetite, enhances satiety, and increases metabolic rate to induce dramatic weight loss. Clinical trials demonstrate greater improvements than single-hormone treatments.
How much weight can people expect to lose with retatrutide?
Trials cite significant average weight loss, frequently more than 20 percent of body mass across months. Individual outcomes differ with dosage, compliance, and initial health. Ask your clinician for individualized expectations.
Who is an ideal candidate for retatrutide treatment?
They’re seeking ideal candidates among adults with obesity (BMI of 30 or higher) or those who are severely overweight (BMI of 27 or higher) with weight-related conditions. A medical evaluation should verify appropriateness, notably for individuals with diabetes, heart disease, or other chronic conditions.
What is the typical treatment protocol for retatrutide?
Protocols in trials employed weekly subcutaneous injections with slow dose escalation across months along with lifestyle counseling. Precise dosing and treatment length await further studies and approvals.
What are the common side effects and risks?
Common side effects include gastrointestinal symptoms, such as nausea, vomiting, diarrhea, and constipation. Less common risks can include gallbladder problems, heart rate alterations, or hypoglycemia in combination with other diabetes medications. Medical supervision is a must.
Can retatrutide replace diet and exercise?
No. Retatrutide supplements, not supplants, diet and exercise. Medications and behavior changes lead to better outcomes that are more sustainable and healthier.
When will retatrutide be widely available and approved?
Retatrutide remains in clinical trials and regulatory review. Access will be subject to trial outcomes and approvals in each country. Consult with a medical professional regarding immediate access and options.